Scientific Computing and Data / Research Data Services / Data Ark: Data Commons / Mount Sinai COVID-19 Biobank
Mount Sinai COVID-19 Biobank

Overview
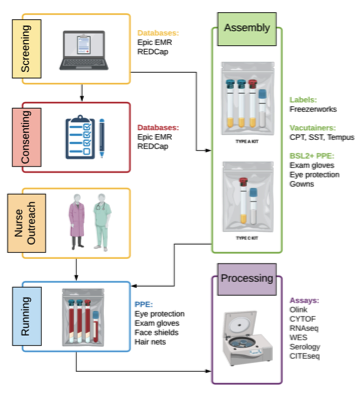
The Mount Sinai COVID-19 Biobank is a joint effort between the Mount Sinai Clinical Intelligence Center (MSCIC) and Human Immune Monitoring Center (HIMC) that arose organically during the COVID-19 pandemic to rapidly collect blood samples from hundreds of COVID-19 patients admitted to Mount Sinai hospitals and serve as the backbone for COVID-19 research. During the peak of the first surge, a volunteer team of more than 100 research staff, nurses, doctors, and scientists collected biological specimens from nearly 800 individuals and collection has continued into 2021. The core team behind the biobank has generated a diverse set of molecular data using state-of-the-art technologies in immunology and genomics and used that data to gain valuable insight into COVID-19. Now, in collaboration with the Office of Research Services, MSCIC and HIMC are releasing the COVID-19 biobank data through the Data Ark to approved applicants in an effort to further expand COVID-19 research within Mount Sinai.
Access
To use this data, you must read, agree and sign the Data Use Agreement (you must be logged in through the Mount Sinai campus network or secure remote VPN). On Minerva, you can load module $ module load dataark to see the path variables.
More Information
If you have questions about the data, please contact Steven Ascolillo at steven.ascolillo@mssm.edu
If you are interested in more information or would like to collaborate with the Mount Sinai Clinical Intelligence Center, please reach out to mscicinfo@mssm.edu
To find out more about the Human Immune Monitoring Center, please click here.
Data Ark Data Sets
Please visit the Data Ark Data Set webpage to explore other data sets.
