CCCEH, CREW, CADRE cohorts:
Our work concentrates on the mechanisms for the onset of asthma and other immunological disorders. One large research focus involves establishing and studying a birth cohort from Northern Manhattan (Columbia Center for Children’s Environmental Health, CCCEH), determining the importance of environmental allergens, traffic-related pollutants, and phthalate exposure to the onset of allergies, asthma, and allergic immune responses. A major emphasis is on the role of prenatal and early postnatal exposure on later childhood and adolescent asthma risk. Additional areas of research include identifying novel genetic by environment interactions and epigenetic by environment interactions important to the onset of asthma.
Subsequently, we teamed up with investigators from 12 asthma birth cohorts across the U.S. to establish the Children’s Respiratory Research Workgroup (CREW) consortium. CREW proposed to identify specific types of childhood asthma, develop an understanding of what early life environmental influences cause these different types of asthma and when, and identify targets for future efforts aimed at preventing childhood asthma. CREW included data from a large number of children (over 9,000 at birth) and their families, with broad diversity in terms of ethnicity, family characteristics, neighborhoods and geographic locations. One of the primary goals of CREW is to put together sets of data and samples of participating cohorts to identify phenotypes of childhood asthma (i.e. specific subtypes of asthma that can be distinguished by clinical features such as natural history, triggers, exacerbation frequency, concurrent allergies, lung function, sex, etc). We are obtaining mechanistic insights about personal and early life risk factors, and connecting asthma phenotypes with underlying causes and pathogenic mechanisms to define endotypes of childhood asthma. More recently, CREW has expanded into the Children’s Allergy and Asthma Data Repository” (CADRE) consortium.
Children’s Respiratory Research Workgroup (CREW) and Children’s Allergy and Asthma Data Repository (CADRE) cohorts. 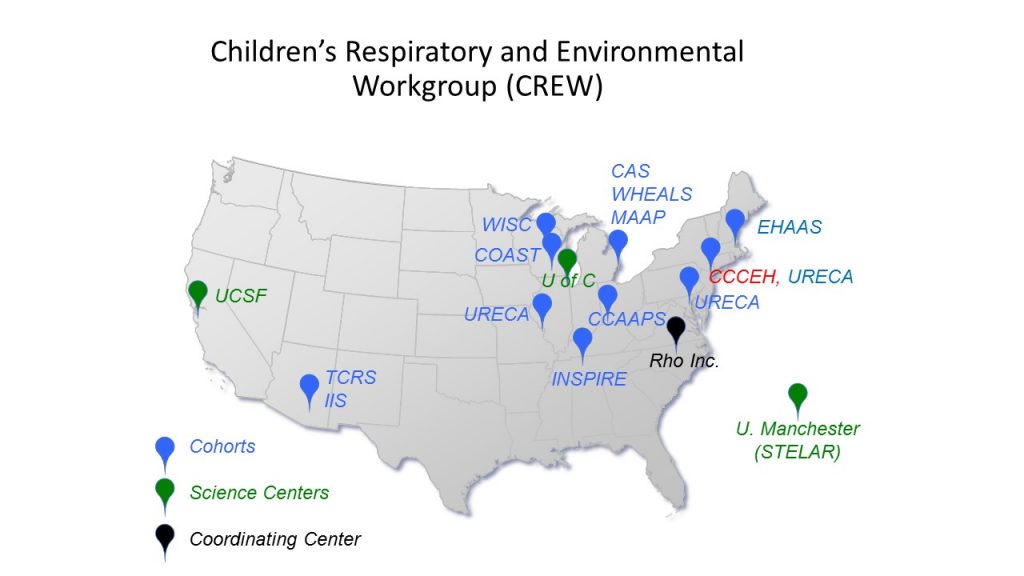
Our individual CCCEH study site also participates in the larger NIH Environmental Influences on Child Health Outcomes (ECHO) program. Dr. Miller co-leads the research derived from the NYC cohort study site. The Miller lab has led published papers on the role of chemicals in childhood asthma and allergic disorders and on risk factors for the onset of childhood asthma with recurrent exacerbations. The overall goal is to help generate science and identify actionable solutions to prevent disease and promote better health among children.
Endocrine Disruption, Hormones, and Sex Differences in Adolescent Airway Health:
This research leverages past findings from a well-retained multi-center cohort (TIDES. Representing San Francisco, CA; Minneapolis, MN; Rochester, NY; and Seattle, WA) to determine relationships between prenatal and childhood phthalates and phenols (including novel replacement chemicals), serum sex steroid hormones (estrogens and testosterone), targeted urinary steroid metabolites, and sex differences in airway health through adolescence. In the co-investigator role, Dr. Miller and her team have been applying expertise in endocrine disrupting chemical exposure and childhood asthma and advising on all aspects of this research. This includes helping with the design of the exposure assessment, analytical plan and interpretation of results. She oversees the training in the measurement of fractional exhaled nitric oxide and the conduction of spirometry at all sites. She has been responsible for ensuring proper quality controls and assurances of these data and overreads all the spirometry for this proposal.
Biologics And Clinical Immunology Cohort at Sinai (BACICS):
Many chronic immune mediated disorders (IMDs) are increasing in prevalence and continuing to cause substantial morbidity and mortality. Despite the development of biologics that precisely target pathological immune pathways, clinical responses remain uneven. The reasons for this variability remain a foremost research gap. Emerging data suggest that environmental exposures may explain why some patients exhibit more immune resilience whereas others experience persistent immune dysregulation. We launched the Biologics And Clinical Immunology Cohort at Sinai (BACICS) that is comprised of chronic IMD patients to apply state-of-the-art approaches towards assessing clinical and immunological patterns of treatment responses that transcend previous single disease and static paradigms. The cohort is designed to determine mechanisms for immune resilience longitudinally, including those that occur in common response to environmental exposures and those that pertain to multiple IMDs. All patients undergo the same repeated environmental, geospatial, nutritional and social assessments with comprehensive biobanking. The new cohort focuses on shared features of systemic inflammation and immune dysregulation. This contrasts to previously applied approaches that focused on individual diseases and may not have captured sufficiently the common environmental causes or the symptoms that result from these shared immune pathologies.
