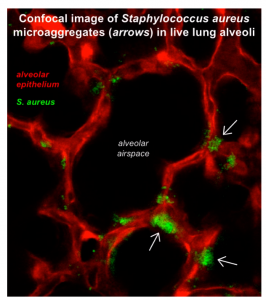
Staphylococcus aureus, a major lung pathogen, rapidly forms microaggregates in structural niches of lung alveoli to stabilize, initiate alveolar infection, induce alveolar damage, and resist antibiotic therapy.
This project focuses on mechanisms by which inhaled bacteria infect and damage lung alveoli (PMID 29431734). We discovered that contact with the alveolar epithelium causes inhaled S. aureus bacteria (green fluorescence in confocal microscopic image above) to form microaggregates (arrows) in structural niches of the alveolar wall (red fluorescence). Microaggregate formation stabilizes the bacteria in the protected niche locations, leading to secretion of bacterial toxins, damage to the niche alveolar wall, and spread of damage signals through alveolar cell-cell communication. The result is widespread fluid leakage into the air spaces, respiratory failure, and death. Since microaggregate formation protects bacteria from antibiotics, coupling antibiotic therapy with microaggregate dispersal approaches increases antibiotic efficacy and survival. A video version of the abstract can be viewed here.
The novelty of these findings center on a new role for alveolar microanatomy in lung disease pathogenesis and drug efficacy. Importantly, this project challenges the notion that the extensive spatial profile of bacteria-induced lung injury is caused by widespread lung infection or extensive activation of the immune system. Rather, our findings bring to light a new concept, that lung injury rapidly results from bacterial toxin-induced damage to niche-localized alveolar walls, which is amplified to healthy lung by cell-to-cell spread of the damage signals. This concept explains the clinical observation that localized S. aureus lung infection can quickly progress to severe pneumonia, respiratory failure, and death.
This paper was published in 2018 in the Journal of Clinical Investigation and highlighted by the editors in “JCI This Month”. Related abstracts have garnered five awards and eleven oral presentations at national and international scientific meetings. The abstract submitted to the 2018 meeting of the American Thoracic Society was selected from a pool of 700 abstracts to receive a Science and Innovation Center Award.
Ongoing Research Projects
Our ongoing research builds on our prior work. Funded projects focus on the endogenous mechanisms by which S. aureus-damaged alveoli are repaired, and alveolar responses to influenza that promote secondary alveolar infection by S. aureus, leading to fatal influenza-staphylococcal coinfection. Since lung infection disproportionately affects the young and the old, aspects of our ongoing work aim to address how alveolar responses to inhaled bacteria and viruses change across the lifespan.
