Research
The goal of our research is to better understand the effects of chronic metabolic diseases, such as diabetes, on brain function. People with diabetes have double the rate of mental illness compared to those who don’t have diabetes and are twice as likely to not respond to current treatments. Currently, we use mouse models of diabetes to identify the ways in which chronic hyperglycemia can lead to behaviors associated with mental illness by exploring processes at the behavioral, circuit, cellular and molecular levels. A major focus of our work is on hyperglycemia-induced changes in gene expression within cholinergic neurons in the brain’s reward circuitry. We also use operant models to explore how diabetes affects learning and response to rewards. In the near future, we plan to repeat many of these behavioral studies in people with diabetes or at risk of developing diabetes.
PubMed publications
https://www.ncbi.nlm.nih.gov/pubmed/?term=ables+jl
Google scholar
https://scholar.google.com/citations?user=w9gNODEAAAAJ&hl=en

Assistant Professor
Annenberg 19-38
Phone: 212-241-2774
jessica.ables@mssm.edu
Funding and Awards
Funding:


![]()
![]()
![]()
NIDA R01DA059455
Awards:
Jessica Ables receives a Travel Award for the Molecular Psychiatry meeting. (November 2023)
Jessica Ables receives a Travel Award for the annual meeting of the Society for Biological Psychiatry. (April 2023)
Jessica Ables is awarded a Pilot & Feasibility Award from the Einstein-Mount Sinai Diabetes Research Center. (September 2022)
Jessica Ables is awarded a Whitehall Foundation Research Grant. (December 2021)
Jessica Ables is awarded an Alkermes Pathways Research Award. (July 2021)
Jessica Ables is awarded a NARSAD Young Investigator Grant. (October 2019)
Jessica Ables is elected to the Alpha Omega Alpha Honor Society. (June 2019)
Jessica Ables wins Best Poster, 2nd Place, Friedman Brain Institute Neuroscience Retreat. (May 2019)
Jessica Ables is elected to the Career Development Institute for Psychiatry Class of 2019. (January 2019)
Jessica Ables is named Laughlin Fellow, American College of Psychiatrists. (January 2019)
Jessica Ables is named a Leon Levy Fellow in Neuroscience. (April 2018)
Jessica Ables is named Psychiatry Chief Resident for Research. (2018)
Jessica Ables receives the NIMH Outstanding Resident Award. (July 2017)
Jessica Ables is named as a Junior Mentor for the Icahn School of Medicine MSTP. (2017)
Projects
1. Nitric oxide in development of tolerance
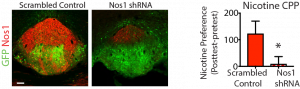
The interpeduncular nucleus (IPN) is a GABAergic nucleus that is characterized by high expression of neuronal nitric oxide synthase (NOS1) as well as striking vascularity. We have found that NOS1 is increased in IPN after chronic exposure to nicotine, and more recently, to oxycodone. We hypothesize that this increase in NOS1 mediates the development of tolerance to both the aversive and positive effects of drugs. Our goal is to determine if this increase in NOS1 in the IPN generalizes to all other drugs of abuse and might represent a target for the general treatment of addiction. Future studies will focus on visualizing nitric oxide release and the effects of nitric oxide on the epigenome, transcriptome and function of neurons in the reward circuitry.
2. Cholinergic vulnerability to hyperglycemia
 Animal models of diabetes indicate that cholinergic neurons are particularly vulnerable to persistent hyperglycemia and resulting oxidative and nitrosative stress, and we hypothesize that this vulnerability may contribute to the neurocognitive and affective impairments observed in diabetic patients. The medial habenula (MHb) is a highly conserved cholinergic nucleus in the epithalamus that projects to the IPN to regulate levels of serotonin, dopamine and epinephrine and, as a result, mood. The MHb is susceptible to degeneration after repeated administration of drugs of abuse, however, to date no one has examined the effect of persistent hyperglycemia on the functional or structural plasticity of the MHb. Our goal is to understand the molecular, cellular and functional consequences of persistent hyperglycemia on cholinergic neurons in the reward circuitry using cell-type specific epigenomic and transcriptional profiling in combination with viral-mediated gene transfer. We will then relate these changes to behavior.
Animal models of diabetes indicate that cholinergic neurons are particularly vulnerable to persistent hyperglycemia and resulting oxidative and nitrosative stress, and we hypothesize that this vulnerability may contribute to the neurocognitive and affective impairments observed in diabetic patients. The medial habenula (MHb) is a highly conserved cholinergic nucleus in the epithalamus that projects to the IPN to regulate levels of serotonin, dopamine and epinephrine and, as a result, mood. The MHb is susceptible to degeneration after repeated administration of drugs of abuse, however, to date no one has examined the effect of persistent hyperglycemia on the functional or structural plasticity of the MHb. Our goal is to understand the molecular, cellular and functional consequences of persistent hyperglycemia on cholinergic neurons in the reward circuitry using cell-type specific epigenomic and transcriptional profiling in combination with viral-mediated gene transfer. We will then relate these changes to behavior.
Check out our recent publication examining mitochondrial morphology within the MHb in a mouse model of diabetes.
And take a look at our collaboration with the Sweis lab to examine foraging behavior in a mouse model of diabetes.
3. Axonal transcriptome
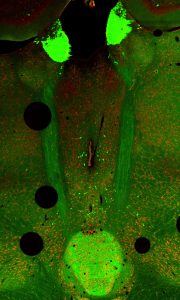 Recent research has revealed that neuronal axons have specialized mechanisms to regulate delivery and utilization of mRNAs independently of the neuronal cell body, where the nucleus resides. This is not surprising given that in many cases (e.g. motor neurons in the limbs), the axons are far removed from the cell body and require the ability to respond to stimuli on timescales that would prohibit gene expression from the nucleus and subsequent delivery to the distal axon. Projection neurons in the brain provide a unique opportunity to profile the transcripts that are utilized in discrete compartments of the cell, as the axons are physically separate from the cell body. Our lab is currently profiling axonal transcriptomes in specific cell populations in the normal mouse brain during fear learning. Our goal is to determine how translation is locally regulated in response to various stimuli, including stress and metabolic derangements.
Recent research has revealed that neuronal axons have specialized mechanisms to regulate delivery and utilization of mRNAs independently of the neuronal cell body, where the nucleus resides. This is not surprising given that in many cases (e.g. motor neurons in the limbs), the axons are far removed from the cell body and require the ability to respond to stimuli on timescales that would prohibit gene expression from the nucleus and subsequent delivery to the distal axon. Projection neurons in the brain provide a unique opportunity to profile the transcripts that are utilized in discrete compartments of the cell, as the axons are physically separate from the cell body. Our lab is currently profiling axonal transcriptomes in specific cell populations in the normal mouse brain during fear learning. Our goal is to determine how translation is locally regulated in response to various stimuli, including stress and metabolic derangements.Current Team

Jessica Ables, MD/PhD
Assistant Professor
jessica.ables@mssm.edu


Eftychia Markopoulou
Research Associate (2025-)
eftychia.markopoulou@mssm.edu

Mohammad Jodeiri Farshbaf, PhD
Postdoctoral Researcher (2021-)
mohammad.jodeirifarshbaf@mssm.edu


Mariam Mahboob
Neuroscience PhD student
mariam.mahboob@icahn.mssm.edu

Natasha Rice
Master’s Student (2023-)
natasha.rice@icahn.mssm.edu

Taelor Matos
PREP Student (2023-)
taelor.matos@icahn.mssm.edu
Current Volunteers
Jake Tetenman – Washington University at St. Louis
Zim Khan – McNair Scholar, Hunter College, CUNY
Hridika Tasnim – McNair Scholar, Hunter College, CUNY
Former Members

Associate Researcher (2018-2022)

University of Minnesota – SURP4US Student (2022) – Now an MSTP student at Icahn School of Medicine at Mount Sinai

Jillian Haller
Scranton University – Undergraduate Volunteer (2022)

Krist Niblo
Master’s Student – (2020-2023)
Former Volunteers
Gabby Chavez (2018-2023) – Pelham Memorial High School & Fordham University
Martha Solomon (2019) – SUNY Downstate Medical Student
Nazeen Shah (2021-2022) -Spackenkill High School
Almer Yu (2022) – Hunter College High School
Yacoub Alokam – St. Francis College Undergraduate
Sophia Leng – Hunter College High School
Alp Vargelci – Stuyvesant High School
Natalia Zheng – Stuyvesant High School
Namrata Marjit, MS – Icahn School of Medicine
Stella Lodewick – Clemson University
Potential Lab Members
Icahn School of Medicine at Mount Sinai is seeking enthusiastic, detail-oriented applicants. The School is one of the nation’s leading medical schools, ranked in the top 20 schools for NIH funding. Both the Psychiatry and Neuroscience Departments offer an outstanding intellectual and multidisciplinary research environment with a commitment to translational research. Note that we do not offer observerships for foreign medical graduates.
Volunteers:
We typically accept 1-2 high school and 1-2 undergraduate volunteers each year, with the majority of the time in the lab spent during the summer. Volunteers are expected to work independently with supervision after training. Interested candidates should send their resume/CV in January for a summer position.
Graduate Students:
All graduate students interested in our lab must first apply and be accepted into a program at Mount Sinai.
Postdoctoral Fellows:
Postdoctoral candidates should have a Ph.D. and/or M.D. and a strong basic science background in molecular/cellular neuroscience. Those with experience in the following fields are particularly encouraged to apply: computational approaches for analysis of sequencing datasets, machine learning approaches for animal behavior, electrophysiology, fiber photometry or in vivo imaging.
Interested candidates should submit their Curriculum Vitae and contact information for at least three professional references to Jessica.ables@mssm.edu.
ANNENBERG BUILDING 19-38
Icahn School of Medicine at Mount Sinai
1 Gustave L. Levy Pl, New York, NY 10029
