Molecular and Cellular Mechanisms of Neurological Disorders and Therapeutic Development
The Yue Laboratory focuses on investigating the molecular, cellular, network, circuitry, and behavioral mechanisms underlying various neurological disorders and psychiatric conditions, including Parkinson’s disease, Alzheimer’s disease, Huntington’s disease, bipolar disorder, and schizophrenia. The Yue lab employs a multidisciplinary approach, utilizing experimental and bioinformatic methods to gain insights into the pathogenesis of these disorders and to develop potential therapeutic strategies.
1. Disease Mechanisms: We focus on cellular processes such as autophagy-lysosome degradation, synaptic vesicle trafficking and activity, cellular metabolism, neuroinflammation, and protein/lipid kinase signaling.
2. Experimental Techniques: Mouse genetics, biochemical assays, molecular and cellular biology approaches, primary neuron/glia cultures, cellular imaging (both live and fixed), transcriptomics (including single-cell RNA sequencing and spatial transcriptomics), proteomics, bioinformatics, and animal pathology/behavioral studies.
3. Human-Relevant Models: Human-induced pluripotent stem cell (iPSC)-derived neurons and glia (2D and 3D), postmortem tissue analysis
4. Translational Research: Therapeutic target identification and validation, disease modification development focused on nanobody, gene therapy, and drug repositioning.
Pathogenic mechanism of neuron and glia interactions in Parkinson’s disease
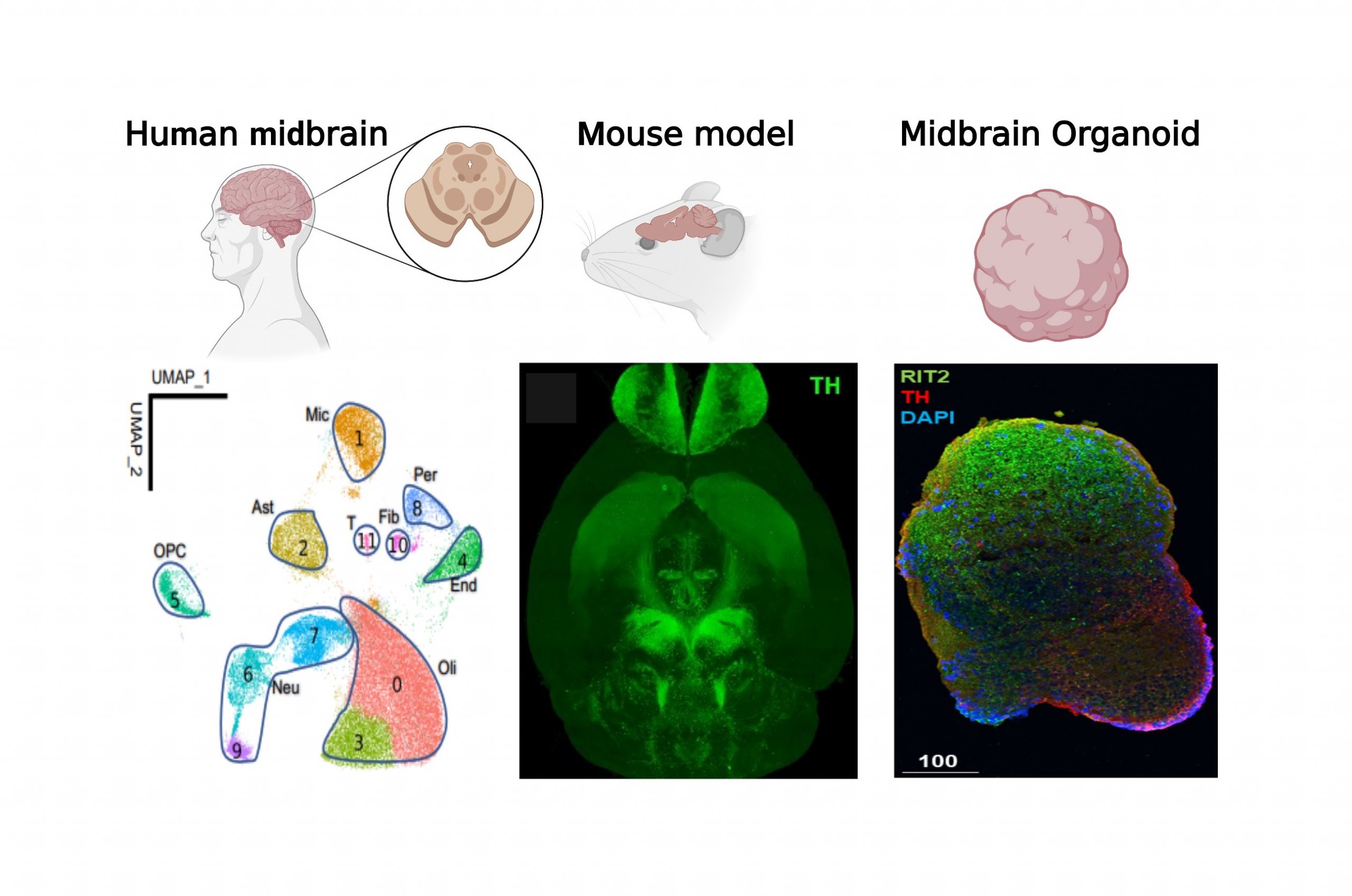
We are undertaking the profiling and characterization of molecularly distinct subtypes of neurons and glia in the substantia nigra pars compacta (SNpc) from Parkinson’s disease (PD) brains. Our goal is to identify novel cell types and unravel the molecular mechanisms underlying the vulnerability and resilience of neurons in PD. We employ a multidisciplinary approach, integrating network modeling with experimental validation using human postmortem tissues, genetic mouse models, and induced human neurons (iPSC) in both 2D and 3D midbrain cultures. We aim to model and characterize human dopamine neuron subpopulations, pinpointing those associated with PD vulnerability within the SNpc, utilizing hiPSC-based models. See our PD Center website: https://icahn.mssm.edu/research/center-parkinsons-disease
Molecular mechanism of body-brain interactions during the progression of Parkinson’s disease
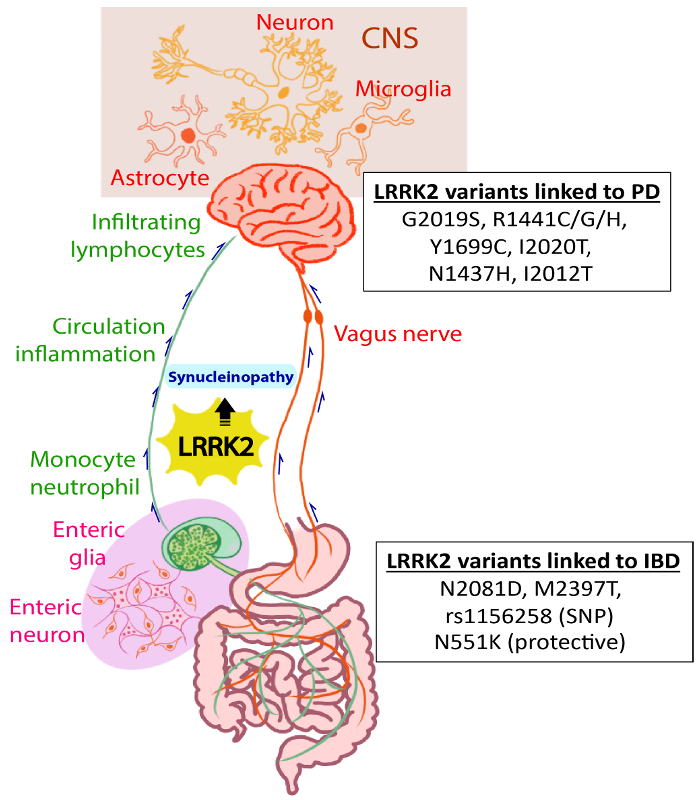
An increased incidence of PD in patients with inflammatory bowel disease (IBD), a chronic disease of the gut, has also been reported world-wide. With the finding of alpha-synuclein-associated Lewy body in peripheral tissues (e.g. gut) of PD patients, there is a growing appreciation for the hypothesis of gut-brain axis and chronic inflammation in the disease onset and progression for PD. The link between intestinal inflammation and PD is also enforced by elevated markers of intestinal inflammation. We aim to determine the subtypes of PD associated with prodromal gut chronic inflammation, dissect the pathogenic mechanism underpinning gut-brain axis, and identify molecular biomarkers and novel therapeutics for subtypes of PD. We will test the hypothesis that (1) chronic inflammation in the gastrointestinal system contributes to a subset of PD, and this is mediated by pathogenic interaction between LRRK2 and alpha-synuclein; (2) LRRK2 acts as an interface for the signaling pathways that leads to PD and IBD, and investigation of LRRK2 targets (e.g. small GTPase Rab proteins) will help elucidate the molecular mechanisms for the gut-brain axis in the pathogenesis of PD.
Microglial autophagy and cellular senescence in Alzheimer’s disease and Parkinson’s disease
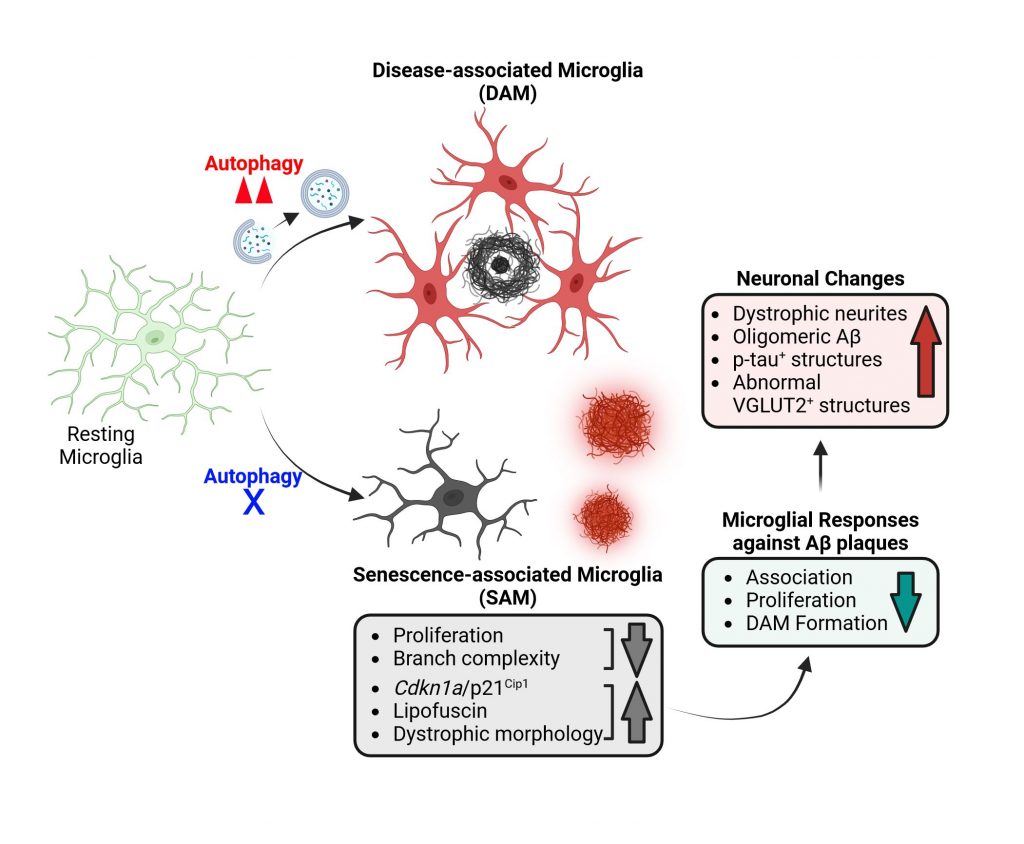
Autophagy represents an evolutionarily conserved lysosomal degradation system crucial for cellular homeostasis. Its regulation varies across cell types and is intricately linked to the pathogenic pathways of neurodegenerative diseases. Our research endeavors aim to elucidate the neuroprotective mechanisms conferred by autophagy in microglia and to comprehend how dysfunctional autophagy in microglia contributes to cellular senescence and the pathogenesis of Alzheimer’s disease (AD) and Parkinson’s disease (PD). Our central hypothesis posits that cellular senescence is accelerated in microglia in the AD and PD brains. Through our investigations, we seek to delineate the role of microglial autophagy in modulating inflammation and curtailing senescence via selective degradation processes. Our ultimate goal is to devise therapeutic strategies geared towards the removal of senescent microglia to foster neuroprotection during both aging and the progression of neurodegenerative diseases.
Autophagy-lysosome dysfunctions in neurons and glial cells in the pathogenesis of neurodegenerative diseases
We propose to elucidate the cell-type-specific autophagy-lysosome pathways, focusing on neurons and microglia, under both physiological (young and old) and pathological conditions (Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease). Employing a systematic approach, we will incorporate multiple genetic mutant lines of mice, human-induced neuron lines (hiN), quantitative proteomics, and molecular and cell biology techniques to identify autophagy cargo and receptors in neurons and microglia. Furthermore, we will investigate the distinct mechanisms responsible for the degradation of multiple disease-associated proteins such as tau, alpha-synuclein (aSyn), and huntingtin. The findings of our study hold the potential to uncover novel biomarkers and therapeutic targets for autophagy-lysosomes in the treatment of Alzheimer’s, Parkinson’s, and Huntington’s diseases, thereby paving the way for innovative therapeutic interventions.
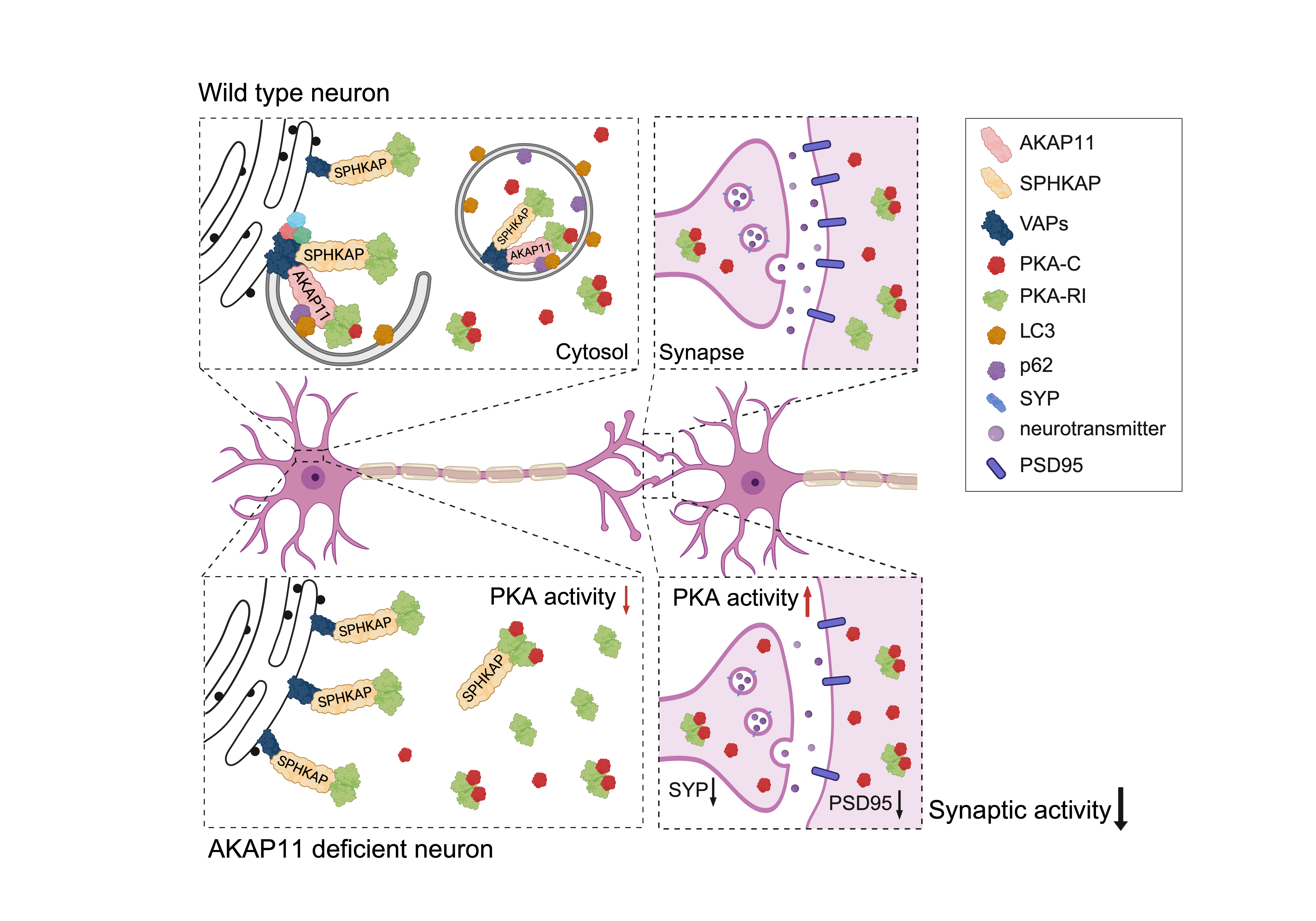
Dysfunctional AKAP11-PKA Signaling Underlies the Mechanism for Synaptic Impairment in Human Psychiatric Disorders (Bipolar and Schizophrenia)
Human genomic studies have identified protein-truncating variants in AKAP11 associated with both bipolar disorder (BD) and schizophrenia (SCZ), implicating a shared disease mechanism driven by loss-of-function in AKAP11. We discovered that AKAP11, a protein kinase A (PKA) adaptor, plays a key role in degrading the PKA-RI complex through selective autophagy. Through multi-omics approaches, cell biology, and electrophysiology analysis in mouse models and human induced neurons, we delineated a central role of AKAP11 in coupling PKA kinase network regulation to synaptic transmission. Loss of AKAP11 distorted compartment-specific PKA and GSK3 activities and impaired cellular functions that significantly overlap with pathways associated with BD and SCZ. Moreover, we identified interactions between AKAP11, the PKA-RI adaptor SPHKAP, and the ER-resident autophagy-related proteins VAPA/B, which co-adapt and mediate PKA-RI complex degradation in neurons. Notably, AKAP11 deficiency impaired neurotransmission, providing key insights into the mechanism underlying AKAP11-associated psychiatric diseases. We are currently interrogating the causality between synaptic PKA hyperactivity and synaptic impairment and testing synaptic PKA activity as a potential drug target for BD and SCZ.

Zhenyu Yue, PhD
*Professor of Neurology and Neuroscience
*Alex and Shirley Aidekman Family Professorship
*Director, Center for Parkinson’s disease Neurobiology
*Director, Basic and Translational Research of Movement Disorder
Biography
https://icahn.mssm.edu/profiles/zhenyu-yue
***
email:
zhenyu.yue@mssm.edu
News
- On May 13th, Q&A video of Dr. Zhenyu Yue and Sharon’s son, George is live. Episode 270 – Investigating the Cellular and Molecular Mechanism Underlying Neurodegenerative Diseases – Parkinson’s (Autophagy & α-Synuclein) is live. Check out here.
- On May 11th & 12th, 2026, Congratulations to Sanem, Nitika, and Ayris on successfully defending their master’s dissertation!
- On May 8th, 2026, Congratulations to Xianjian Li on winning the poster presentation award at the 18th FBI Neuroscience Retreat!
- On May 8th, 2026, Dr. Yue and lab members participated in the 18th Friedman Brain Institute Retreat. You-Kyung, Xianjian, Cong, and Ayris presented their posters. Check out their abstracts here: https://neuroscience.mssm.edu/pdf/Retreat-2026-Abstracts.pdf
- On May 7, 2026, Congratulations to Takahiro, Sanem, and Nitika on their publication in Translational Neurodegeneration
- On May 5, 2026, the Yue lab celebrated Bik Tzu‘s PhD Commencement. We’re so proud of you!
- On April 26th, 2026, Congratulations to Xingjian‘s paper published in npj Parkinson’s Disease.
- On Nov 28th, 2025, Congratulations to You-Kyung Lee and Cong Xiao’s paper published in Nature Communication.
- From Nov 15th to Nov 19th, 2025,Linh Chu, Marianna Liang and Eileen Li presented their research at the SfN, San Diego
- On Oct 30th, 2025, Congratulation to Emily Birnbaum on review paper published in Autophagy Reports
- On August 15th, 2025, Congratulations to Linh Chu on receiving Trainee Professional Development Award from the Society for Neuroscience (SfN)
- On August 6th, Dr. Yue had a Q&A with Sharon’s son, George to share his research in Investigating the Cellular and Molecular Mechanism Underlying Neurodegenerative Diseases-Parkinson’s (Autophagy & α-Synuclein). Check out the podcast here.
- On July 17th, 2025, Congratulations to Marianna on being selected to present a poster at the Regeneron Science to Medicine Forum
- On February 24th, 2025, Welcome to the Yue lab, Emily Birnbaum. Emily has started her postdoctoral training in the Yue Lab.
- On February 12th, 2025, Welcome to the Yue lab, Ayris Izmirli. Ayris has started her dissertation research in the Yue Lab.
- On January 15th, 2025, Congratulations on Marianna and Linh’s recent scientific perspective paper published in Movement Disorder.
- On November 22th, 2024, Congratulations to Marianna on being awarded the F31 Ruth L Kirstein NIH predoctoral fellowship!
- On November 6th, 2024 Dr. Yue gave a presentation titled “Neuronal and Glial Autophagy, Selective Eater and Neuroprotection” at the 97th Annual Meeting of the Japanese Biochemical Society, University of Tokyo, Japan https://pub.confit.atlas.jp/en/event/jbs2024/presentation/1S04m-03
- On October 31st, 2024, we bid a farewell to Dr. George Heaton and Dr. Edward (Ted) Wickstead! Congratulations to the next stage of your career! You will be greatly missed!
- From Oct 5th to 9th, 2024, Dr. Yue and Marianna Liang presented their research at the SFN https://www.sfn.org/-/media/SfN/Documents/NEW-SfN/Meetings/Neuroscience-2024/Abstracts-and-Sessions/Abstract-PDFs/SFN24_Abstracts-PDF-Posters_TUES_AM.pdf
- On October 4th, 2024, Welcome to the Yue lab, Nitika Kamath. Nitika has started her dissertation research in the Yue Lab
- On September 29th, 2024 we congratulate Xingjian on his paper published in Nature Communications. (here!)
- On September 16th, 2024, Welcome to the Yue lab, Sanem Isik. Sanem has started her dissertation research in the Yue lab.
- On August 23rd, 2024, we celebrated the 20th anniversary of the Yue Laboratory at Mount Sinai (8/1/2004 – 8/1/2024). We would like to extend our heartfelt gratitude to all the alumni and current members of the Yue Lab for your hard work and dedication. This milestone would not have been possible without your invaluable contributions.
- On August 22nd, 2024, we bid farewell to Dr. Insup Choi celebrating his contributions in the Yue Lab and wishing him the best as he embarks on a new chapter in his career as a tenure-track faculty position at Northwestern University Feinberg School of Medicine in Chicago. Dr. Choi has been an invaluable member to our team, and we are excited to see the great work he will continue to do in his own laboratory. All the best, Dr. Choi!
- On August 20th, 2024, Dr. Zhenyu Yue shared insights into his research on the cellular and molecular mechanisms underlying Parkinson’s Disease, focusing on the work examining the regulation of autophagy-lysosomal pathway. Check the The MJFF podcast series – Episode 21 here.
- On August 12th and August 19th, 2024, our summer students (Katharine Tang, Ryan Mah, Vivian Zhang, Eileen Li, Alex Lu) gave their end-of-summer presentations. Thank you to all students for your participation in the 2024 Yue Lab Summer Research Program. We are all very impressed with your work!
- On April 26th, 2024, Dr. Yue and lab members participated in The 16th Friedman Brain Institute @2024 Neuroscience Retreat https://friedmanbrain.icahn.mssm.edu/retreat-23/
- On April 18th, 2024, Dr. Yue delivered a research seminar titled “The Landscape of Autophagy in the Brain, Neurological Diseases, and Beyond” at the Center for Translational Medicine and Pharmacology, Mount Sinai.
- On April 12th, 2024, Henry Kim, who has been with us all this time, leaves our lab as he enters medical school. Congratulations on your achievement, we wish you all the best!
- On April 10th, 2024, we extend our heartfelt congratulations to Xiaoting for publishing her paper in Nature Communications. (here!)
- On April, 2024, Welcome to the Yue Lab, Takahiro Shimizu. Taka has embarked on his post-doctoral research journey within the Yue laboratory.
- Dr. Yue and Insup Choi‘s work at Mount Sinai Press (here)!
- From November 10th to 15th, 2023, Dr. Yue, Insup Choi, Xingjian Li, Bik Tzu Huang, Henry Kim, Marianna Liang, and Linh Chu, gave a presentation on their research at the SFN Conference.https://www.sfn.org/meetings/neuroscience-2023/registration
- On November 15th, 2023, congratulations to Linh Chu for her achievement in being awarded the Trainee Professional Development Award by the Society for Neuroscience (SfN).
- On November 2023, Congratulations on George Heaton’s grant from the New York Community Trust.
- On May 26th, 2023 Congratulations to Xiaoting on her successful Ph.D. thesis defense.
- On May 25th, 2023, Congratulations on Insup Choi’s paper published in Nature Cell Biology.https://www.nature.com/articles/s41556-023-01158-0
- On May 12th, 2023, Dr. Yue and a lab member participated in The Friedman Brain Institute @2023 Neuroscience Retreat https://friedmanbrain.icahn.mssm.edu/retreat-23/
- On May 1st, 2023, Congratulations on Insup Choi‘s K01 grant from (NIA/NIH)
- On April 21st, 2023, Welcome to Yue lab, Linh Chu. Linh has started her dissertation research in Yue lab and Zhang Lab
- From March 26th to 29th, 2023 Dr. Yue, Insup Choi, You-Kyung Lee, and Xiaoting Zhou presented their research at the @Keystone Conference Autophagy and Neurodegeneration: Mechanisms to Therapies https://www.keystonesymposia.org/conferences/conference-listing/meeting?eventid=6812
- View our blog on the Parkinson’s Foundation Website! Link: Newest Parkinson’s Foundation Research Center Explores Dopamine Nerve Diversity in Parkinson’s | Parkinson’s Foundation
- On November, 2022 Bik Tzu and Marianna presented their research (poster session) at the SfN meeting.
- Congratulations on Insup Choi’s recent review paper published in Science Advances.
- Congratulations to Dr. Yue and his team for having a $2 Million Research Center Grant from the Parkinson’s Foundation (title: Profiling and Elucidating Molecular Mechanism of Neurodegeneration in Parkinson’s disease) https://bit.ly/3yjbG86
- On Sep 22nd, 2022, Congratulations to Bik Tzu Huang on receiving Trainee Professional Development Award from the Society for Neuroscience (SfN)
- On Sep 9th, 2022, Insup Choi presented his study “Investigation of senescent microglia on Alzheimer’s disease” at the REC seminar of ADRC
- On July 1st, 2022, Congratulations to Insup Choi. Insup has been promoted to Assistant Professor in Yue lab
- On July 1st, 2022, Congratulations to George Heaton on winning the best poster award in LRRK2 conference
- On June 27th, 2022, Welcome to Yue lab, Ted Wickstead. Ted has started his post-doctoral research in Yue lab
- On May 27th, 2022, Congratulations to Bik Tzu on becoming a T32 trainee by the Department of Pharmacological Science
- On May 13th, 2022, Congratulations to Insup Choi on winning the oral presentation award at the 14th FBI Neuroscience Retreat
- On Mar 7th, 2022, Welcome to Yue lab, Marianna Liang. Marianna has started her dissertation research in Yue lab
- On Jan 26th, 2022, Congratulations to Ravi Ghotra on his successful graduate thesis defense
- On Jan 10th, 2022, Congratulations to Dr. Insup Choi on winning the ADRC grant (Alzheimer Disease Research Center of Mount Sinai)
- On Nov 1st, 2021, Welcome to Yue lab, Heesoo Kim has joined Yue lab as Associate Researcher
- On Aug 23rd, 2021, Welcome to Yue lab, You-Kyung Lee has started her post-doctoral research in Yue lab
- On May 17th, 2021, Welcome to Yue lab, Bik Tzu Huang. Bik Tzu Huang has started her dissertation research in Yue lab
- On May 13th, 2021, Congratulations to Shiyi on the completion of his training in Yue lab and heading back to China for his thesis defense
- On May 10th, 2021, Congratulations to Steven Seegobin on his admission to LECOM medical school!!!
- On Apr 2nd, 2021, Researchers in Yue lab have found a novel therapeutic target for specific cancer treatment, here is the details
- On Jan 27th, 2021, Lab member Steven Seegobin gave a talk titled “Maintenance of Alpha-Synuclein Homeostasis by Microglia in Parkinson’s Disease” at the ADND/Loeb WIP seminar.
- On Jan 23rd, 2021, Congratulations to Dongxiao on her successful doctorate defense, you well deserved it, Dr. Liang.
- On Jul 1st, 2020, the latest update on Autophagy’s role by Prof. Yue, here are the details
- On Jun 2nd, 2020, Congratulations to Steven Seegobin, his PD animal review paper was accepted by Front Neuroscience, here are the details
- On Oct 1st, 2020, Congratulations to George Heaton!, Dr. Heaton has won first place in 2020 Poster Presentation Award at Morris K. Udall Centers of Excellence for Parkinson’s disease research.
- On Sep 1st, 2019, Congratulations to Insup Choi! Dr. Choi has won first place in 2019 Poster Presentation Award at Morris K. Udall Centers of Excellence for Parkinson’s disease research.
- On Nov 1st, 2011, Pioneering Research of Autophagy at Yue Lab
- On Mar 25th, 2011, Mount Sinai Researchers Uncover How a Gene Mutation Causes Parkinson’s Disease.
Open Positions
Post-Doctoral Fellow – Neurodegenerative Disease’s Mechanisms (PD, AD, and HD) (multiple positions)
The laboratory of Professor Zhenyu Yue is looking for highly motivated and collaborative postdoctoral fellows to join Friedman Brain Institute at Icahn School of Medicine at Mount Sinai in New York City.
Multiple positions are available for highly motivated and ambitious scientists to use a wide range of experimental and bioinformatic approaches to investigate the pathogenic mechanisms of neurodegenerative diseases such as Parkinson’s, Alzheimer’s and Huntington’s diseases. The lab employs mouse genetics, biochemical, molecular, cellular, transcriptomic (snRNAseq), proteomic, network, imaging (live and fixed), animal pathology/behavior, human iPSC neurons/glia and postmortem tissue analysis to dissect autophagy-lysosome, synaptic vesicle trafficking, cell metabolism, neuroinflammation, and protein/lipid kinase signaling in the neuropathogenesis.
Qualifications:
- A recent Ph.D. graduate with demonstrated experience in any of the following fields: cell biology, molecular biology, mouse genetics, biochemistry, neuroscience, and/or systems biology.
- The ideal candidates will have experience in any of the following research areas: molecular and cell biology in neuronal and/or glial culture models; confocal and/or stereological microscopy; protein biochemistry, proteomics; viral vector technology; development of genetic animal models; rodent neuropathology and behavior; bioinformatic, single cell/nuclei RNAseq, iPSC derived neurons/glia,
Please submit your online application with the following:
- Cover letter outlining research experience and interests
- Curriculum Vitae
- Contact details for 3 references
Questions may be directed to Dr. Zhenyu Yue (zhenyu.yue@mssm.edu)
2026
- The emerging role and therapeutic targeting of autophagy-lysosome pathway in the pathogenesis of Parkinson’s disease. Takahiro Shimizu, Sanem Isik, Nitika Kamath, Zhenyu Yue. Transl Neurodegener 15, 21 (2026). https://doi.org/10.1186/s40035-026-00555-3
- Disruption of the LRRK2 substrate RAB12 facilitates neurotransmission and causes hyperactivity in mice. Xingjian Li*, Yuanxin Chen*, Huaixing Wang*, Xue Zhang, Noah Guy Lewis Guiberson, Xianting Li, Jacqueline Burré, Junmin Peng, Hui Zhang, and Zhenyu Yue. npj Parkinson’s Disease (2026).
2025
- Bipolar and schizophrenia risk gene AKAP11 encodes an autophagy receptor coupling the regulation of PKA kinase network homeostasis to synaptic transmission
You-Kyung Lee*, Cong Xiao*, Xiaoting Zhou, Le Wang, Meghan G. McReynolds, Zhiping Wu,Xian Han, Eric Purisic, Henry Kim, Xianting Li, Zhiping Pang, Jinye Dai, Junmin Peng, Nan Yang, Zhenyu Yue. Nature Communications. - Autophagy in the Central Nervous System: Homeostatic Role and Neurological Diseases
Henry Kim*, Edward S Wickstead*, Xiaoting Zhou, Insup Choi, Zhenyu Yue. Nature Neuroscience (in press). - The role of autophagy in synucleinopathy: clearance versus spread of α-synuclein. Emily Birnbaum, Zhenyu Yue. Autophagy Reports. 2025 Oct 30. 10.1080/27694127.2025.2577406
- Targeting Specific Kinase Substrates Rescues Increased Colitis Severity Induced by the Crohn’s Disease-Linked LRRK2-N2081D Variant. George R. Heaton, Xianting Li, Xingjian Li, Xiaoting Zhou, Yuanxi Zhang, Duc Tung Vu, Marc Oeller, Ozge Karayel, Quyen Q. Hoang, Meltem Ece Kars, Nitika Kamath, Minghui Wang, Leonid Tarassishin, Matthias Mann, Inga Peter, Zhenyu Yue. J of Clinical Investigation. 2025 Oct 1. doi: 10.1172/JCI190017
- WSTF nuclear autophagy regulates chronic but not acute inflammation. Yu Wang, Vinay V. Eapen, Yaosi Liang, Athanasios Kournoutis, Marc Samuel Sherman, Yanxin Xu, Angelique Onorati, Xianting Li, Xiaoting Zhou, Kathleen E. Corey, Kuo Du, Ana Maria Cabral Burkard, Chia-Kang Ho, Jing Xie, Hui Zhang, Raquel Maeso-Díaz, Xinyi Ma, Ulrike Rieprecht, Murat Cetinbas, Lu Wang, Jihe Liu, Corey Bretz, Zhuo Zhou, Shannan J. Ho Sui, Srinivas Vinod Saladi, Ruslan I. Sadreyev, Peter D. Adams, Robert E. Kingston, Anna Mae Diehl, Benjamin Alman, Wolfram Goessling, Zhenyu Yue, Xiao-Fan Wang*, Terje Johansen*, Zhixun Dou*. Nature. 2025 Jul 2. doi: 10.1038/s41586-025-09234-1. PMID: 40604282
- New Multiomics Studies Shed Light on Cellular Diversity and Neuronal Susceptibility in Parkinson’s Disease. Marianna Liang*, Linh Chu*, 2025 Jan 15.
2024
-
RAB12-LRRK2 complex suppresses primary ciliogenesis and regulates centrosome homeostasis in astrocytes. Xingjian Li, Hanwen Zhu, Bik Tzu Huang, Xianting Li, Heesoo Kim, Haiyan Tan, Yuanxi Zhang, Insup Choi, Junming Peng, Pingyi Xu, Ji Sun, 2024 Sep 29;15(1):8434.
- Integrated proteomics reveals autophagy landscape and an autophagy receptor controlling PKA-RI complex homeostasis in neurons. 2024 Apr 10;15(1):3113.doi: 10.1038/s41467-024-47440-z.
- Silencing Parkinson’s risk allele Rit2 sex-specifically compromises motor function and dopamine neuron viability. 2024 Feb 23;10(1):41.
- Molecular profiling of human substantia nigra identifies diverse neuron types associated with vulnerability in Parkinson’s disease.2024 Jan 12;10(2):eadi8287.
2022
-
Regulation of α-Synuclein Homeostasis and Inflammasome Activation by Microglial Autophagy. Insup Choi, George R. Heaton, You-Kyung Lee, and Zhenyu Yue (2022) Science Advances. Oct 28;8(43):eabn1298. doi: 10.1126/sciadv.abn1298.
-
Pharmacological modulation of autophagy for Alzheimer’s disease therapy: Opportunities and obstacles. Deng Z, Dong Y, Zhou X, Lu JH, Yue Z. Acta Pharm Sin B. 2022 Apr;12(4):1688-1706. doi: 10.1016/j.apsb.2021.12.009. Epub 2021 Dec 18.PMID: 35847516 Free PMC article. Review.
- Enhancing autophagy maturation with CCZ1-MON1A complex alleviates neuropathology and memory defects in Alzheimer disease models.Cai CZ, Zhuang XX, Zhu Q, Wu MY, Su H, Wang XJ, Iyaswamy A, Yue Z, Wang Q, Zhang B, Xue Y, Tan J, Li M, He H, Lu JH. Theranostics. 2022 Jan 24;12(4):1738-1755. doi: 10.7150/thno.64148. eCollection 2022.PMID: 35198070
2023
2021
- Selective autophagy of AKAP11 activates cAMP/PKA to fuel mitochondrial metabolism and tumor cell growth. Proc Natl Acad Sci.
- Wang Q, Zhang B, and Yue Z (2021). Disentangling the Molecular Pathways of Parkinson’s Disease using Multiscale Network Modeling Trends in Neuroscience
- Wang M, Li A, Sekiya M, Beckmann ND, Quan X, Schrode N, Fernando MB, Yu A, Zhu L, Cao J, Lyu L, Horgusluoglu E, Wang Q, Guo L, Wang YS, Neff R, Song WM, Wang E, Shen Q, Zhou X, Ming C, Ho SM, Vatansever S, Kaniskan U, Jin J, Zhou MM, Ando K, Ho L, Slesinger PA, Yue Z, Zhu J, Katsel P, Gandy S, Ehrlich ME, Fossati V, Noggle S, Cai D, Haroutunian V, Iijima KM, Schadt E, Brennand KJ, and Zhang B (2021). Transformative Network Modeling of Multi-omics Data Reveals Detailed Circuits, Key Regulators, and Potential Therapeutics for Alzheimer’s Disease. Neuron.
2020
- (2020). Autophagy and Parkinson’s Disease. Adv Exp Med Biol.
- Zhao Y, Qin L, Pan H, Liu Z, Jiang L, He Y, Zeng Q, Zhou X, Zhou X, Zhou Y, Fang Z, Wang Z, Xiang Y, Yang H, Wang Y, Zhang K, Zhang R, He R, Zhou X, Zhou Z, Yang N, Liang D, Chen J, Zhang X, Zhou Y, Liu H, Deng P, Xu K, Xu K, Zhou C, Zhong J, Xu Q, Sun Q, Li B, Zhao G, Wang T, Chen L, Shang H, Liu W, Chan P, Xue Z, Wang Q, Guo L, Wang X, Xu C, Zhang Z, Chen T, Lei L, Zhang H, Wang C, Tan J, Yan X, Shen L, Jiang H, Zhang Z, Hu Z, Xia K, Yue Z, Li J, Guo J, Tang B (2020). The Role of Genetics in Parkinson’s Disease: A Large Cohort Study in Chinese Mainland Population Brain.
- Wu JH, Li YN, Chen AQ, Hong CD, Zhou YF, Li PC, Y, Mao L, Xia YP, He QW, Jin HJ, Yue Z, and Hu B (2020). Inhibition of Sema4D/PlexinB1 signaling alleviates vascular dysfunction in diabetic retinopathy. EMBO Molecular Medicine.
- Choi I, Zhang Y, Seegobin SP, Pruvost M, Wang Q, Purtell K, Zhang B & Yue Z (2020). Microglia clear neuron-released α-synuclein via selective autophagy and prevent neurodegeneration. Nature Communications.
2019
- Lachance V, Wang Q, Sweet E, Choi I, Cai CZ, Zhuang XX, Zhang Y, Jiang JL, Blitzer RD, Bozdagi-Gunal O, Zhang B, Lu JH, Yue Z (2019). Autophagy protein NRBF2 has reduced expression in Alzheimer’s brains and modulates memory and amyloid-beta homeostasis in mice. Molecular Neurodegeneration.
- Wang Q, Zhang Y, Wang M, Song WM, Shen Q, McKenzie A, Choi I, Zhou X, Pan PY, Yue Z*, Zhang B* (2019). The landscape of multiscale transcriptomic networks and key regulators in Parkinson’s disease. Nature Communications.
- Schaffner A, Li X, Gomez-Llorente Y, Leandrou E, Memou A, Clemente N, Yao C, Afsari F, Zhi L, Pan N, Morohashi K, Hua X, Zhou MM, Wang C, Zhang H, Chen SG, Elliott CJ, Rideout H, Ubarretxena-Belandia I, Yue Z (2019). Vitamin B12 modulates Parkinson’s disease LRRK2 kinase activity through allosteric regulation and confers neuroprotection. Cell Research.
2018
- Lee YA, Noon LA, Lee T, Akat KM, Chou HI, Berres ML, Fiel MI, Gordon R, Pfleger C, Germain D, Khambu B, Yin XM, Campbell K, Yue Z, Kramer E, John G, Czaja MJ, Hoshida Y and Friedman SL(2018). Autophagy is a Gatekeeper of Hepatic Differentiation and Carcinogenesis by Controlling the Degradation of Yap. Nature communications.
- Yang N and Yue Z(2018). A Peek of Parkinson’s Disease Progression through Human Dopamine Neuron in a Dish. Trends in Neuroscience.
- Hui KY, Fernandez-Hernandez H, Hu J, Schaffner A, Pankratz N, Hsu NY, Chuang LS, Carmi S, Villaverde N, Li X, Rivas M, Levine AP, Bao X, Labrias PR, Haritunians T, Ruane D, Gettler K, Chen E, Li D, Schiff ER, Pontikos N, Barzilai N, Brant SR, Bressman S, Cheifetz AS, Clark LN, Daly MJ, Desnick RJ, Duerr RH, Katz S, Lencz T, Myers RH, Ostrer H, Ozelius L, Payami H, Peter Y, Rioux JD, Segal AW, Scott WK, Silverberg MS, Vance JM, Ubarretxena-Belandia I, Foroud T, Atzmon G, Pe’er I, Ioannou Y, P. B. McGovern D, Yue Z, Schadt EE, Cho JH, and Peter I (2018). Functional variants in the LRRK2 gene confer shared effects on risk for Crohn’s disease and Parkinson’s disease. Science Translational Medicine.
- Wang C, Kang X, Zhou L, Chai Z, Wu Q, Huang R, Xu H, Hu M, Sun X, Sun S, Li J, Jiao R, Zuo P, Zheng L, Yue Z, and Zhou Z (2018). Synaptotagmin-11 is a critical mediator of parkin-linked neurotoxicity and Parkinson’s disease-like pathology. Nature Communications.
- Wu S, He Y, Yang W, Liu W, Li X, Qiu X, Li Y, Shen HM, Wang R, Yue Z, and Zhao Y (2018). Targeting the potent Beclin 1-UVRAG coiled-coil interaction with designed peptides enhances autophagy and endolysosomal trafficking. Proceedings of the National Academy of Sciences.
2017
- Deng Z, Purtell K, Lachance V, Wold M, Chen S, and Yue Z (2017). Autophagy receptors and Neurodegeneration. Trends in Cell Biology.
2016
- Guaitoli G, Raimondi F, Gilsbach BK, Gómez-Llorente Y, Deyaert E, Renzi F, Li X, Schaffner A, Jagtap PKA, Boldt K, Zweydorf FV, Gotthardt K, Lorimer DD, Yue Z, Burgin A, Janjic N, Sattler M, Versées W, Ueffing M, Ubarretxena-Belandia I, Kortholt A and Gloeckner CJ(2016). A structural model of the Parkinson’s Disease protein LRRK2 provides insight into the intramolecular regulation of its kinase activity. Proceedings of the National Academy of Sciences.
2015
- Lim J, Yue Z (2015). Neuronal Aggregates: Formation, Clearance, and Spreading. Developmental Cell.
2014
- Lu J, He L, Behrends C, Araki M, Araki K, Wang QJ, Catanzaro JM, Friedman SL, Zong WX, Fiel MI, Li M, Yue Z (2014). NRBF2 regulates autophagy and prevents liver injury by modulating Atg14L-linked phosphatidylinositol-3 kinase III activity. Nature communications.
- Yamamoto A and Yue Z (2014). Autophagy and its Normal and Pathogenic States in the Brain. Annual Reviews of Neuroscience.
- Tang G, Gudsnuk K, Kuo SH, Cotrina ML, Rosoklija G, Sosunov A, Sonders MS, Kanter E, Castagna C, Yamamoto A, Yue Z, Arancio O, Peterson BS, Champagne F, Dwork AJ, Goldman J, Sulzer D (2014). Loss of mTOR-Dependent Macroautophagy Causes Autistic-like Synaptic Pruning Deficits. Neuron.
- Friedman AK, Walsh JJ, Juarez B, Ku SM, Chaudhury D, Wang J, Li X, Dietz DM, Pan N, Vialou VF, Neve RL, Yue Z, Han MH (2014). Enhancing depression mechanisms in midbrain dopamine neurons achieves homeostatic resilience. Science.
2013
- Yue Z and Yang XW (2013). Dangerous duet: LRRK2 and alpha-synuclein jam at CMA. Nature Neuroscience.
- Yang Y, Coleman M, Zhang L, Zheng X, Yue Z (2013). Autophagy in axonal and dendritic degeneration. Trends in Neuroscience.
2012
- Friedman L, Lachenmayer L, Wang J, He L, Poulose S, Komatsu M, Holstein G, Yue Z (2012). Disrupted autophagy leads to dopaminergic axon and dendrite degeneration and promotes presynaptic accumulation of a-synuclein and LRRK2 in the brain. The Journal of Neuroscience.
- Steele JW, Lachenmayer ML, Ju S, Stock A, Liken J, Kim SH, Delgado LM, Alfaro IE, Bernales S, Verdile G, Bharadwaj P, Gupta V, Barr R, Friss A, Dolios G, Wang R, Ringe D, Fraser P, Westaway D, St George-Hyslop PH, Szabo P, Relkin NR, Buxbaum JD, Glabe CG, Protter AA, Martins RN, Ehrlich ME, Petsko GA, Yue Z, Gandy S (2012). Latrepirdine improves cognition and arrests progression of neuropathology in an Alzheimer’s mouse model. Molecular Psychiatry.
- Li X, He L, Che KH, Funderburk SF, Pan L, Pan N, Zhang M, Yue Z*, Zhao Y* (2012). Imperfect interface of Beclin1 coiled-coil domain regulates homodimer and heterodimer formation with Atg14L and UVRAG. Nature Communications.
- Sheng Z, Zhang S, Butos D, Kleinheinz T, Le Pichon CE, Domingues S, Solanoy HO, Drummond J, Zhang X, Ding X, Li X, Yue Z, Liu X, Burkick DJ, Estrada AA, Gunzner-Toste J, Chen H, Sweeney ZK, Sceace-Levie K, Moffat JG, Kirkpatick DS, Zhu H (2012). Autophosphorylation of Serine on LRRK2 is an in vivo biomarker of kinase activity and is required for LRRK2-induced neuronal toxicity. Science Translational Medicine.
2010-2002
- Funderburk SF, Wang QJ, and Yue Z (2010). The Beclin 1-VPS34 complex – at the crossroads of autophagy and beyond. Trends in Cell Biology.
- Li X, Patel JC, Wang J, Avshalumov MV, Nicholson C, Buxbaum JD, Elder GA, Rice ME, Yue Z (2010). Enhanced striatal dopamine transmission and motor performance with LRRK2 overexpression in mice is eliminated by familial Parkinson’s disease mutation G2019S. The Journal of Neuroscience.
- Zhong Y, Wang QJ, Li X, Chait BT, Heintz N, and Yue Z (2009). Distinct Regulation of Autophagic Activity by Novel Components Atg14L and Rubicon in Beclin 1-Vps34/phosphatidylinositol (PtdIns) 3-kinase complex. Nature Cell Biology.
- Komatsu M, Waguri S, Koike M, Sou YS, Ueno T, Hara T, Mizushima N, Iwata J, Ezaki J, Murata S, Hamazaki J, Nishito Y, Iemura S, Natsume T, Yanagawa T, Uwayama J, Warabi E, Yoshida H, Ishii T, Kobayashi A, Yamamoto M, Yue Z, Uchiyama Y, Kominami E, Tanaka K (2007). Homeostatic levels of p62 control cytoplasmic inclusion body formation in autophagy-deficient mice. Cell.
- Komatsu M, Wang QJ, Holstein GR, Friedrich VL Jr, Iwata J, Kominami E, Chait BT, Tanaka K, Yue Z (2007). Essential role for autophagy protein Atg7 in the maintenance of axonal homeostasis and the prevention of axonal degeneration. Proceedings of the National Academy of Sciences.
- Wang QJ, Ding Y, Kolhz DS, Mizushima N, Cristea IM, Rout MP, Chait BT, Zhong Y, Heintz N, Yue Z (2006). Induction of autophagy in axonal dystrophy and degeneration. The Journal of Neuroscience.
- Yue Z, Jin S, Yang C, Levine AJ, Heintz N (2003). Beclin1, an autophagy gene essential for early embryonic development, is a haplo-insufficient tumor suppressor. Proceedings of the National Academy of Sciences.
- Yue Z, Horton A, Bravin M, DeJager PL, Selimi F, Heintz N (2002). A Novel Protein Complex linking the d2 Glutamate Receptor (GluRd2) and Autophagy: Implications for Neurodegeneration in Lurcher Mice. Neuron.
Team


Zhenyu Yue, PhD
Aidekman Family Professorship
Professor of Neurology and Neuroscience

Xingjian Li
Postdoctoral Fellow
Research Topic: Parkinson’s disease

You-Kyung Lee
Postdoctoral Fellow
Research Topic: Neurodevelopmental disorder, Neurodegenerative disease






Intern student
Lab Alumni
<Postdoc fellow>
Qing-Jun Wang (2004-2006) -University of Kentucky
Jing Wang (2008-2013) -Private Biotech Company, Cleveland, Ohio,
Liqiang He (2007-2011) -Novartis, NY
Bernadette Saunier- Rebori (2008-2011) -Research Institute scientist
Lenard Lachenmayer (2009-2012) -Switzerland, Doctor
Yun Zhong (2007-2007) -Private investment institute, New York
Sarah Funderburk (2009-2011) -Scientific Associate at International Meetings & Science
James Powell (2012-2015) -Mount Sinai
Nicole McKnight (2012-2015) -Private Company
Shibu Poulose (2005-2007) -United States Department of Agriculture’s Human Nutrition Research Center on Aging at Tufts University
Eric Sweet (2012-2016) -University of West Chester
Junghyun Lim (2013-2017) -Genentech, Senior Scientist
Jiahong Lu (2012-2014) -University of Macao
Qian Wang (2016-2018) -Mount Sinai School of Medicine
Ping-Yue Pan (2014-2018) -Rutgers University
Veronik Lachance (2015-2019) -CIHR Health Research Training Award for postdoctoral fellow
Kerry Purtell (2015-2019) -Crohn’s disease Foundation
Patricia Sheehan (2017-2019) -Private company NIH-F32 training fellowship
Zhiqiang Deng (2015-2020) -Hong Kong Baptist University
Mathilde Pruvost (2016-2018) -CUNY
Catalina Requejo (2018-2019) -Spain
Marian Blanca Ramirez (2019-2020) -Fisher Scientific
Insup Choi (2016 – 2020) – Icahn School of Medicine at Mount Sinai (2020 – 2024)/ University of Northwestern
George Heaton (2020 – 2024) – Alzforum
Edward (Ted) Wickstead (2022 – 2024) – Midwestern University
<PhD & MD/PhD Student>
Lauren Friedman (2007-2012) -Alzheimer’s disease Foundation
Mitchell Wold (2010-2016) -Epion Health
Alexandra Keenan (2016-2016) -Mount Sinai
ShiYi Pan (2017-2020) Wuhan University
Dong-xiao Liang (2018-2020) -Central South University Univ/Xiangya Medical School
Xiaoting Zhou (2019-2023) -Central South University Univ/Xiangya Medical School
<Master & Intern Student>
Trinh Pham
Adam Schaffer (2014-2019) -Mount Sinai School of Medicine
Steven Seegobin (2017-2019) -Mount Sinai
Xiaoluan Hua (2018-2019) -Icahn school of medicine at Mount Sinai
Ravi Ghotra (2020-2022) -Nova Southeastern University School of Medicine
Henry Kim (2021-2024) -MD/PhD student in Rutgers
<Research Assistants>
Heesoo Kim (2022-2024)
Contact Us
We accept rotation students from Mount Sinai PhD, MD-PhD, MD, & MS programs
For postdoc position, please contact
zhenyu.yue@mssm.edu
Mail Address:
One Gustave L. Levy Place
New York, NY 10029
Lab Location
1470 Madison Avenue
Hess Center 9-201, 202 (lab), 9-106 (office)
New York, NY 10029