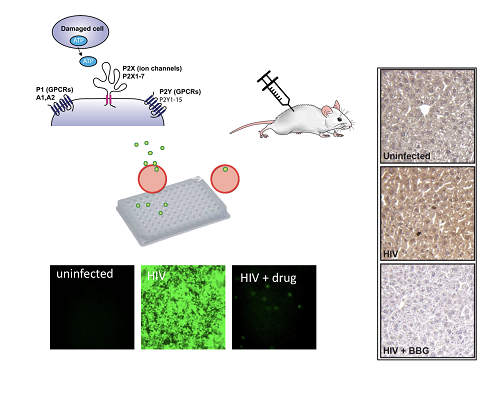
 Targeting purinergic receptors to block HIV infection and inflammation.
Targeting purinergic receptors to block HIV infection and inflammation.
This project is aimed at understanding the mechanism by which purinergic receptors interact with HIV and how antagonists of purinergic receptors block HIV infection. The laboratory has been exploring the role of purinergic signaling in HIV disease and has demonstrated that P2X receptors specifically are required for HIV viral membrane fusion. These studies utilize cell culture, human ex vivo tonsil models, and humanized mice. Cells are infected cells using fluorescence reporter viruses to test the role of purinergic inhibitors and the deletion of P2X genes using CRISPR-Cas9. These studies would be the first of their kind in establishing a new class of drugs for HIV that could serve both to reduce the HIV reservoir and to reduce associated inflammation, thus reducing the burden of chronic HIV disease, and potentially leading to insights into a cure.
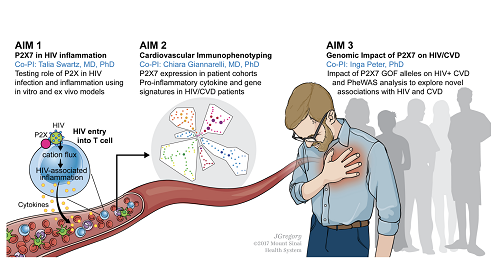 The Role of P2X7 in HIV-Infected Individuals with Atherosclerotic Cardiovascular Disease
The Role of P2X7 in HIV-Infected Individuals with Atherosclerotic Cardiovascular Disease
This multidisciplinary project will bring together expertise in infectious diseases (Dr. Talia Swartz), immunology and cardiovascular disease (Dr. Chiara Giannarelli), and genomics (Dr. Inga Peter) to utilize Core facilities and resources at Mount Sinai to understand the role of P2X7 in HIV and ACVD disease testing P2X7 expression, immune activation, and gene expression. Our goal is to define P2X7 among drivers of inflammation in ACVD in HIV-infected patients in order to develop predictive paradigms and novel therapeutics.
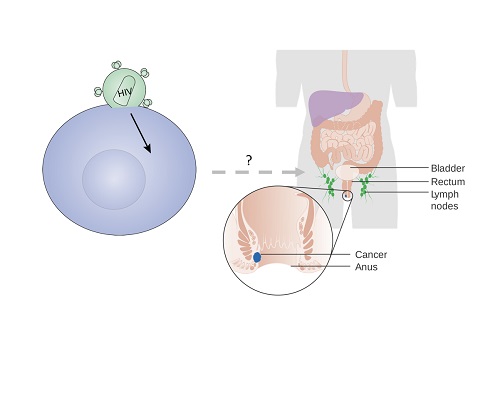 The Role of HIV on the Immune and Inflammatory Microenvironment in Anal Dysplastic Lesions
The Role of HIV on the Immune and Inflammatory Microenvironment in Anal Dysplastic Lesions
This project represents a collaboration with Dr. Keith Sigel at the Icahn School of Medicine Division of General Internal Medicine to determine the impact of immune and inflammatory microenvironment in the natural history and treatment response of anal malignancy in patients infected with HIV. The goal of these studies is to determine whether HIV infection potentiates immune exhaustion and drives inflammatory changes associated with progression of anal cancer. Our approach is to characterize the immune response using banked biopsy specimens from a cohort of HIV infected and uninfected individuals.
