Computational Modeling- DFGmodel
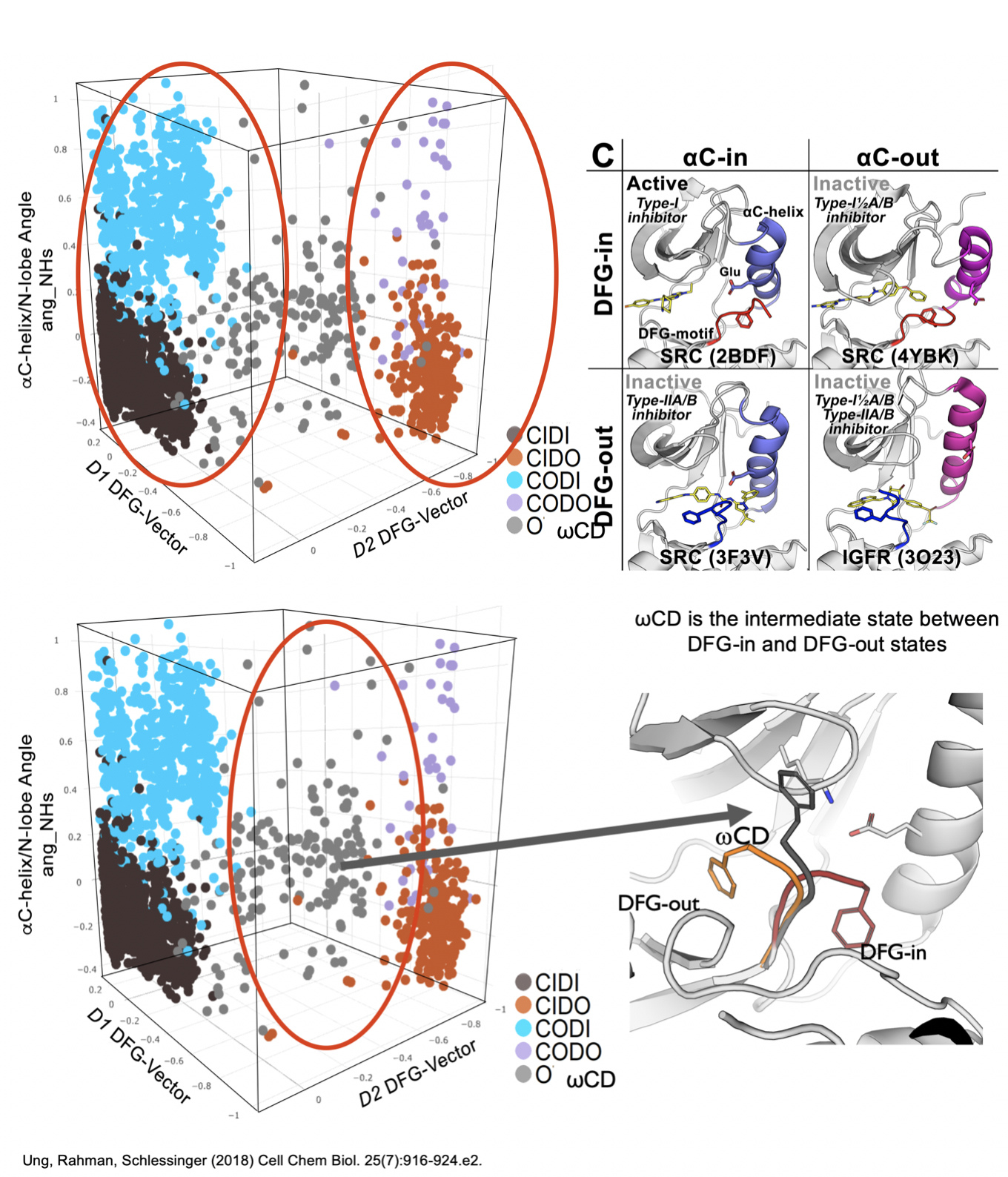
We have introduced computational drug prediction methods into our integrated platform. We use a variety of structural modeling, virtual screening, and cheminformatics tools to screen millions of purchasable and synthesizable compounds into our screening platform. For example, DFGmodel is a recent development by the Schlessinger lab that generates structural models of inactive state of kinases, facilitating compound design of kinase inhibitors of particular types such as the type II kinase inhibitor sorafenib and its analogs. In our most recent collaborative studies we used DFGmodel to guide the design of clinically relevant sorafelogs (Sonoshita et al, NCB 2018) and to develop kinase inhibitors with chemically unique scaffolds (Ung et al, PLOS CB, 2019). We are currently incorporating machine learning algorithms into our pipeline as well as other advanced cheminformatics techniques. For example, we have recently used a random forest algorithm to classify kinase structural states based on a range of structural features derived from key motifs (DFG loop, C-helix). The method, Kinformation, captures the known structural states of kinases and identifies the previously unknown intermediate state ‘wCD’, thereby refining our currently understanding of the kinase conformational continuum. The method is publicly available in Kinametix.com, where it enables a user to download models in inactive states and identify putative chemical substructures that are associated with each conformational type. The substructures are expected to guide the design of novel type II inhibitors.
DREAM Challenge
Our model organism approach highlighted the complexities that emerge in multigenic, whole animal disease models. To address this we turned to multi-targeted therapies. ‘Polypharmacology’, or ‘network-based’ drugs have key advantages for a host of diseases including cancer, and Mendelian diseases. Recent data supports the view that multi-targeting drugs can show increased efficacy, reduced toxicity, and in the case of cancer reduced susceptibility to emergent resistance. However, we are currently limited in our ability to predict chemical structures that are able to directly bind and inhibit multiple targets.
While using model organisms/genetics plus medicinal/computational chemistry to develop lead compounds is a powerful approach, the ability to move from genetics (predict new pro/anti-targets) to structure (predicted by computational chemistry) is still a difficult task. We therefore collaborated with SAGE Bionetworks to develop a new DREAM Challenge. Our goal is to challenge the computational chemistry field to develop more effective computational prediction algorithms.
Challenge Organizers
Scientific Advisory Board:
• Ross Cagan, Icahn School of Medicine at Mt. Sinai, NY
• Avner Schlessinger, Icahn School of Medicine at Mt. Sinai, NY
• Heather Carlson, University of Michigan
• Ruben Abagyan, U of California San Diego
• Justin Guinney, Sage Bionetworks
• Kristen Dang, Sage Bionetworks
Multi-Targeting Drug DREAM Challenge
In this DREAM challenge, we seek new approaches to predicting the structures of candidates that can bind to and inhibit activity of multiple independent targets. Recent genetic and chemical dissection of models of medullary thyroid carcinoma have identified four key targets that together strongly reduce the ability of oncogenic Ret(M918T) to transform cells: RET, SRC, EPH, and FRK; MKNK1 and BRAF were identified as ‘anti-targets’ (Sonoshita et al, submitted). Overall, these polypharmacological approaches represent an exciting alternative to the single-targeted drugs currently being developed.
We asked Challenge participants to use available databases, public and private, to predict compounds that bind multiple targets. Predictions that took new and promising approaches to drug/multi-target binding were tested in in vitro and/or in vivo assays.
Anticipated outcomes include: 1) Novel and re-usable methods for rational design of multi-targeting compounds and 2) a benchmark standard for assessing multi-targeting compounds. Overall, we received ten submissions, with strongly innovative approaches. Based on our in vitro testing of participants’ predicted compounds, we are pleased to announce Zhaoping Xiong, Shanghai Institute of Materia Medica, Chinese Academy of Sciences in Shanghai, China as a winner of the competition.

Relevant References
Integrated computational and Drosophila cancer model platform captures previously unappreciated chemicals perturbing a kinase network. Peter Man-Un Ung, Masahiro Sonoshita , Alex P. Scopton, Arvin C. Dar, Ross L. Cagan, Avner Schlessinger. bioRxiv
KinaMetrix: a web resource to investigate kinase conformations and inhibitor space.
Rahman R, Ung PM, Schlessinger A. Nucleic Acids Res. 2018 Oct 13. doi: 10.1093/nar/gky916. PMID: 30321373
Redefining the Protein Kinase Conformational Space with Machine Learning.
Ung PM, Rahman R, Schlessinger A. Cell Chem Biol. 2018 Jul 19;25(7):916-924.e2. doi: 10.1016/j.chembiol.2018.05.002. Epub 2018 May 31.
PMID: 29861272
Novel selective thiadiazine DYRK1A inhibitor lead scaffold with human pancreatic β-cell proliferation activity. Kumar K, Man-Un Ung P, Wang P, Wang H, Li H, Andrews MK, Stewart AF, Schlessinger A, DeVita RJ. Eur J Med Chem. 2018 Sep 5;157:1005-1016. doi: 10.1016/j.ejmech.2018.08.007. Epub 2018 Aug 22.
PMID: 30170319
Multi-targeting Drug Community Challenge. Schlessinger A, Abagyan R, Carlson HA, Dang KK, Guinney J, Cagan RL. Cell Chem Biol. 2017 Dec 21;24(12):1434-1435. doi: 10.1016/j.chembiol.2017.12.006. No abstract available.
PMID: 29272700
Small molecule stabilization of the KSR inactive state antagonizes oncogenic Ras signalling. Dhawan NS, Scopton AP, Dar AC. Nature. 2016 Sep 1;537(7618):112-116. doi: 10.1038/nature19327. Epub 2016 Aug 24. PMID: 27556948
Prediction of Genetic Interactions Using Machine Learning and Network Properties.
Madhukar NS, Elemento O, Pandey G. Front Bioeng Biotechnol. 2015 Oct 26;3:172. doi: 10.3389/fbioe.2015.00172. eCollection 2015. Review. PMID: 26579514
