Next generation strategies to target oncogenic BRAF signaling
Components of the RAS/RAF/MEK/ERK signaling cascade (MAPK or ERK pathway) are frequently mutated in cancer, with RAS or BRAF mutations driving growth of more than a third of human tumors. Using a variety of biochemical and genetic approaches, we elucidated the structural basis of the unique biochemical properties of RAF inhibitors as the combined outcome of distinct allosteric mechanisms (Figure 1). Our model predicts the biochemical effect of any RAF inhibitor based on its structural properties and the cellular context, and provides a blueprint for the development of rationally designed RAF inhibitors and improved RAF inhibitor-based therapeutic strategies for BRAF-dependent tumors. Our laboratory is currently focused on gaining a detailed understanding of the biologic and biochemical effects of next generation RAF, MEK and ERK inhibitors with the goal of designing more effective therapeutic strategies in cancers driven by oncogenic BRAF signaling.
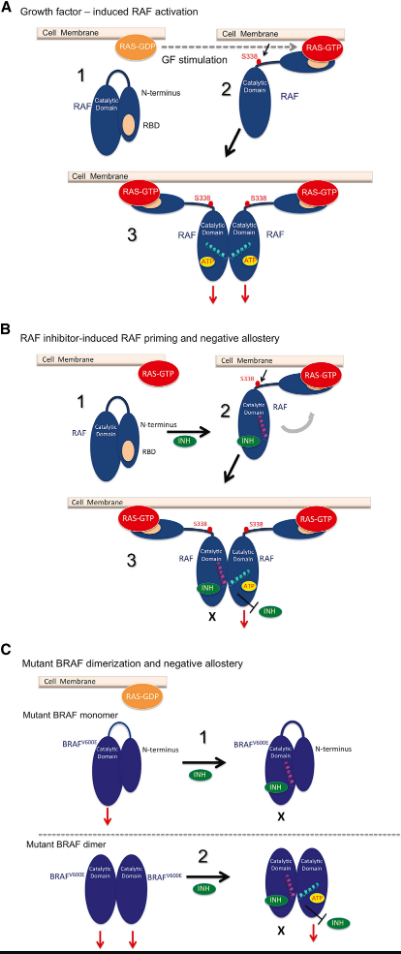
Figure 1
Mechanisms of adaptive resistance to pharmacologic targeting of oncogenic signaling
Adaptive drug resistance is a major challenge to the clinical success of cancer therapies. Incomplete inhibition of oncogenic signaling allows survival of “drug-tolerant” tumor cells, which persist in that state for variable periods before acquiring additional genetic mutations associated with acquired drug-resistance and tumor relapse. We found that combinatorial pharmacologic targeting of ERK signaling and the SHP2 phosphatase prevents adaptive resistance in defined subsets of MAPK-dependent tumors (Figure 2). The strategy was broadly effective in Triple-Negative Breast Cancer models and tumors with RAS mutations at G12, whereas tumors with RAS(G13D) or RAS(Q61X) mutations were resistant. Further, we identified a subset of BRAF(V600E) tumors that were resistant to the combined treatment, in which FGFR was found to drive feedback-induced RAS activation, independently of SHP2. Using a variety of preclinical models and experimental approaches, we are currently investigating the molecular determinants of adaptive resistance to next-generation targeted therapies (RAF, MEK and ERK inhibitors) in MAPK-driven cancers.
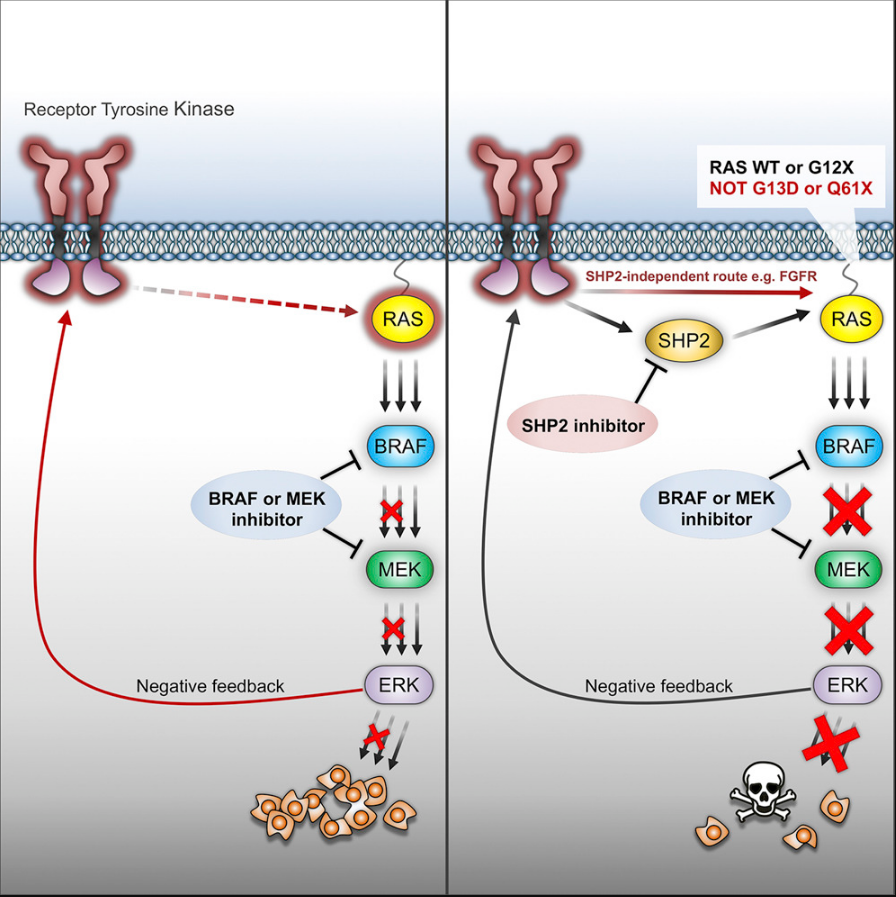
Figure 2
