Finding the Best Combination of Brain and Spinal Cord Stimulation with Hand Training after Spinal Cord Injury
Background
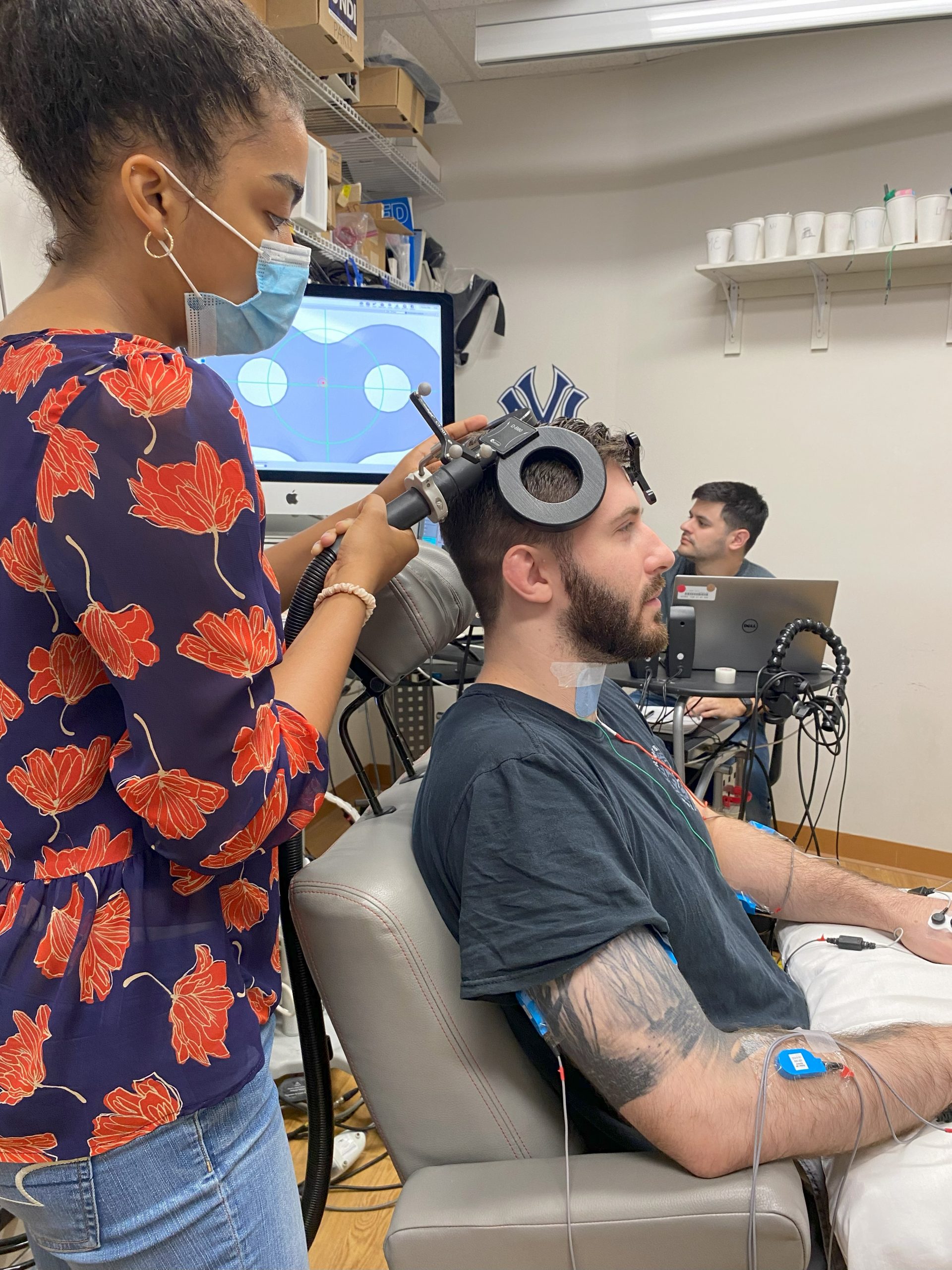
- People with cervical spinal cord injury (SCI) have reduced nerve connections between the brain and the hands. Activating spared nerve connections could be a way to improve recovery.
- We are testing different combinations of magnetic and electrical brain and spinal cord stimulation plus arm/hand exercises to see if we can optimize temporary improvements in hand function for each participant.
- The goal is to determine each participant’s optimal individualized method for delivering repetitive stimulation along with functional hand exercises to obtain a more lasting effect of at least 24 hours.
- Note: This is a research study. This study is testing for temporary changes (up to several days) in nerve transmission and hand function. If we see temporary changes in this study, then future studies would focus on how to prolong that effect.
Eligibility Criteria (summary)
- Age 18-85 years old with chronic cervical SCI, occurred greater than 12 months ago.
- Ability to at least slightly move muscles of left or right hand and fingers.
- No history of severe traumatic brain injury (TBI); not ventilator-dependent; no implanted stimulators or cardiac pacemaker/defibrillator, no epilepsy.
- Other eligibility criteria will be screened in person to make sure it is safe for you to participate
Procedures
- This study involves a significant time commitment: 53 visits over a period of 6-9 months.
- Scheduling is flexible. You do not need to complete all visits.
What you should know
- Electrical and magnetic stimulation can feel like a ‘shock’ that is temporarily uncomfortable at higher pulse strength.
- We have never observed a serious adverse event from study procedures during more than 10 years of stimulation studies.
- If you are uncomfortable at any time for any reason, we can stop the procedures. There is no obligation!!
Investigator
Noam Y. Harel, MD, PhD
Study ID
SCI SCAP+HAND
Contact
Sig Sigafose or Francisco Castano
Clinicaltrials.gov Identifier
NCT06104735