THE TSH RECEPTOR SIGNALING AND SMALL MOLECULES
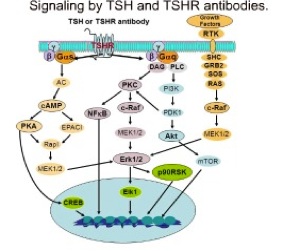
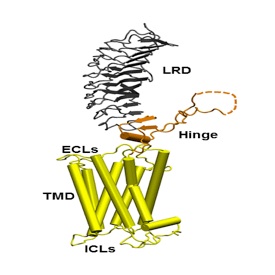
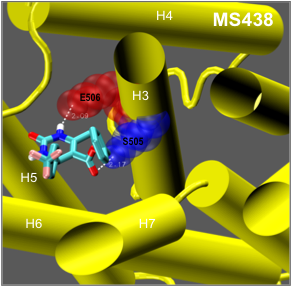
The thyrotropin receptor (TSHR), expressed on the plasma membrane of thyroid epithelial cells, is central to the regulation of thyroid growth and function. The TSHR is also the major autoantigen in autoimmune hyperthyroidism known as Graves’ disease where T cells and autoantibodies are directed at the TSHR antigen. The expressed TSHR undergoes major post-translational changes including (1) intra-molecular cleavage into covalently linked subunits and (2) the formation of dimeric/multimeric forms. Activation of the receptor involves binding of TSH to the large ectodomain of the TSHR followed by interactions between receptor transmembrane domains and G proteins leading to initiation of multiple signaling pathways. This results in thyroid hormone synthesis and secretion as well as cell proliferation and cell survival. Our laboratory was the first to demonstrated the existence of multimeric TSHR forms in native thyroid tissue, and in transfected cells, using functionally tagged TSHRs and fluorescence resonance energy transfer (FRET). Further, using molecular docking studies and high-throughput screening of large chemical libraries we have identified agonists and antagonists against the TSH receptor which is under development as novel therapeutic agents.
ANTIBODIES TO THE TSH RECEPTOR: STRUCTURE-FUNCTION
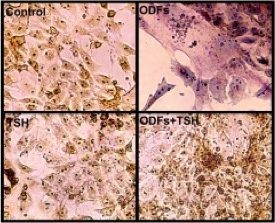
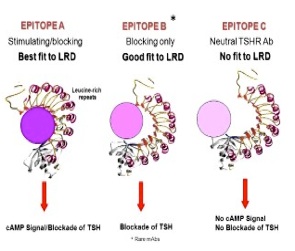
Using a hamster model of hyperthyroid GD developed in our laboratory, we generated a repertoire of hamster monoclonal antibodies (mAbs) to the TSHR with either stimulating, blocking or neutral activity.Our laboratory was the first to publish a high-affinity monoclonal thyroid stimulating antibody (MS-1); at the time this was the “holy grail” of thyroid autoimmunity.We have recently focused deeply on TSHR neutral antibodies and their function. These antibodies on binding to the receptor have no influence on TSH action or receptor activity but we have found that such antibodies can influence other down-stream signaling cascades and also induce stress in thyrocytes. However, we still do not know the full repertoire of TSHR-Abs present in patients or induced in animal models of GD. We know that stimulating TSHR-Abs recognize conformational epitopes on the native TSHR. Therefore, the primary focus of our current studies is to identify the biological functions and signal transduction pathways influenced by TSHR-mAbs.
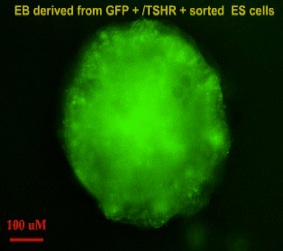
EMBRYONIC STEM CELL MODEL SYSTEMS TO STUDY THYROID DIFFERENTIATION
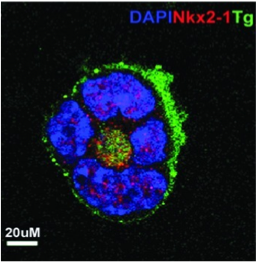
ES cells are continuously growing cell lines isolated from the inner cell mass of the blastocyst that can be propagated indefinitely in an undifferentiated state.Our laboratory uses murine and human ES cells to study thyroid progenitor cell development and differentiation. When ES cells are induced to differentiate in vitro, they form three-dimensional structures called embryoid bodies (EBs) that are composed of derivatives of the three embryonic germ layers and have the potential to differentiate into cells of all lineages. Using the ES/EB differentiation model, we have demonstrated that cultures of EB-derived adherent cell populations can be induced to contain thyrocyte-like cells. We are using these model systems as tools to study novel approaches to develop patient specific IPS cells as a potential for cell replacement therapy in hypothyriodism and also dissect out the molecular and epigentic regulation of thyroid development.
TSH AND BONE
In 2003 our laboratory developed a TSHR knockout mouse in order to explore the extra-thyroidal role of the TSH receptor. The unexpected osteoporotic-like characteristics of the heterozygous and TSHR-KO mouse revealed that TSH has an important modulatory influence on bone remodeling. We have shown that TSHRs are expressed in osteoclasts and osteoblasts, and that one way TSH regulates bone remodeling is by decreasing the production of TNFα in osteoclasts and reducing osteoclastogenesis. Further, our laboratory is focused on studying the role of a TSH and a novel TSH splice-variant that has been discovered in macrophages to understand the role of TSH in the immune-bone axis.