Our Research
The Trivieri Lab focuses on uncovering the molecular mechanisms driving cardiovascular diseases, with an emphasis on heart failure, Cardiomyopathy, Hereditary Hemorrhagic Telangiectasia (HHT), and Pulmonary Hypertension (PH). We use advanced cellular models, tissue engineering systems, and molecular imaging platforms to study disease progression and evaluate therapeutic strategies.
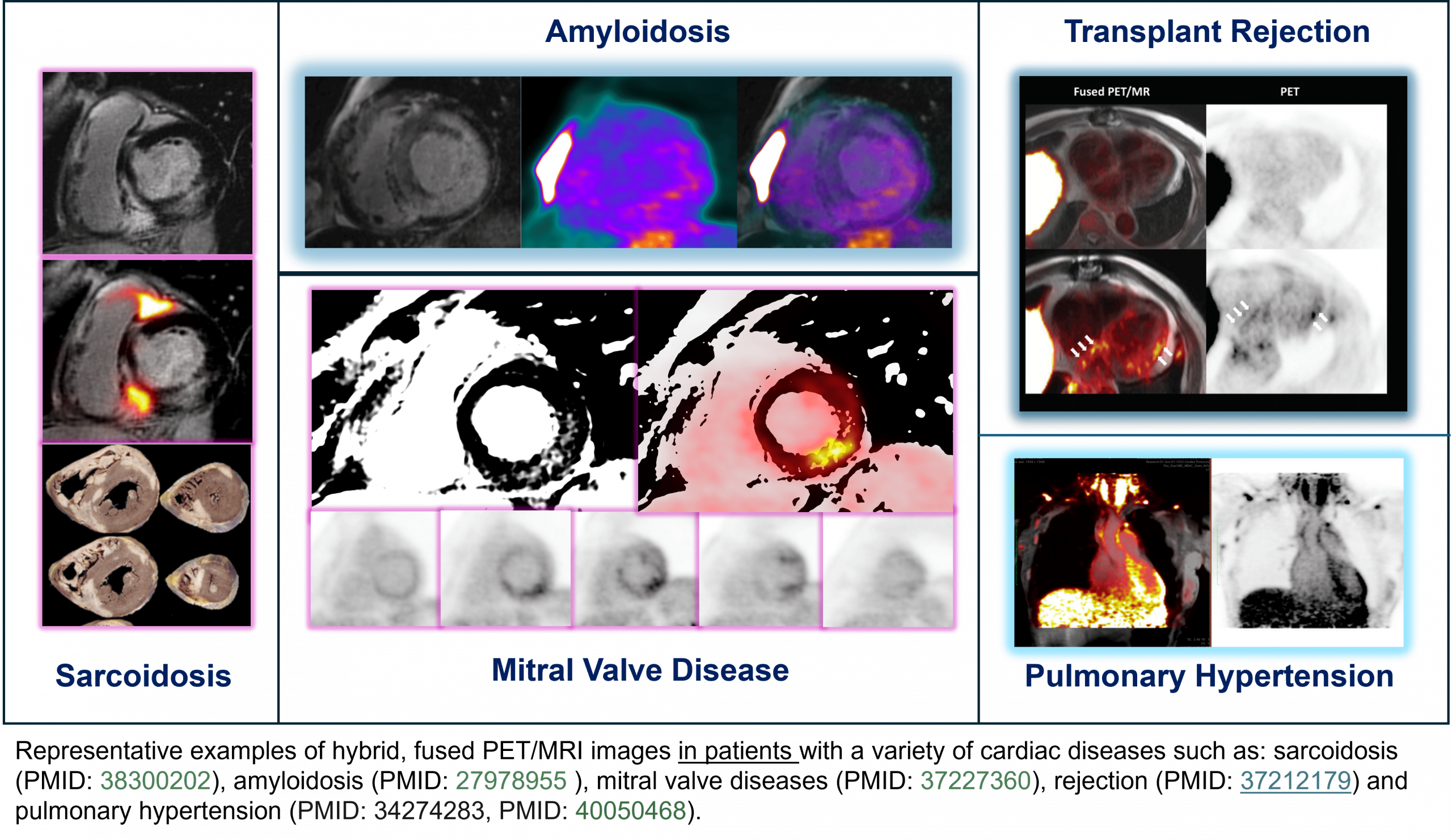
Clinical Imaging Research
As a physician-scientist and active member of the Biomedical Engineering and Imaging Institute (BMII), Dr. Trivieri leads several clinical imaging initiatives that place Mount Sinai at the forefront of PET/MR research in cardiovascular disease. Her work has contributed significantly to characterizing complex cardiac conditions including cardiac sarcoidosis, amyloidosis, mitral valve prolapse, and more recently, COVID-19-associated cardiovascular syndromes.
One particularly groundbreaking study led by Dr. Trivieri uncovered a previously unrecognized link between metabolic abnormalities in the mitral valve and life-threatening arrhythmias. This work was recognized as a key innovation at the Icahn School of Medicine and featured in the Frontiers of Medical Research section of Science magazine.
These efforts have led to multiple successful grant submissions and awards, including collaborations with national and international partners, further advancing translational cardiovascular imaging.
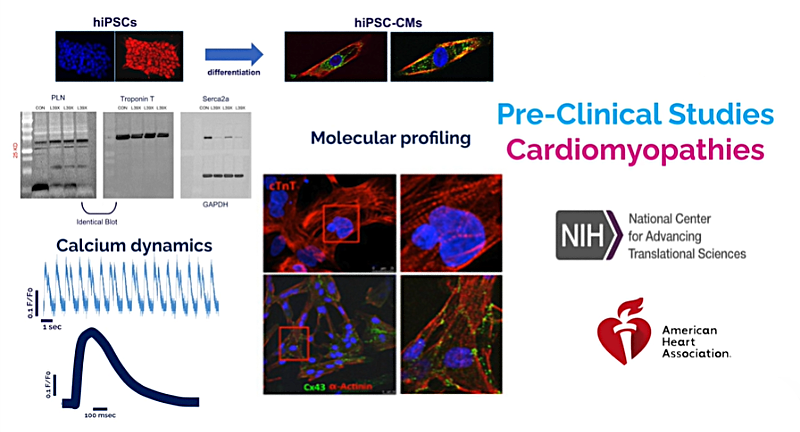
Phospholamban in Cardiomyopathy and Calcium Dysregulation
We investigate the role of phospholamban (PLN) in regulating cardiac function, using patient-derived human iPSCs and mouse models to study cardiomyopathy. Our goal is to understand how calcium dysregulation contributes to heart failure. We apply a wide range of high-throughput and precision techniques, including flow cytometry, mass spectrometry, proteomics, confocal and electron microscopy, as well as engineered cardiac tissues, to uncover druggable molecular mechanisms.
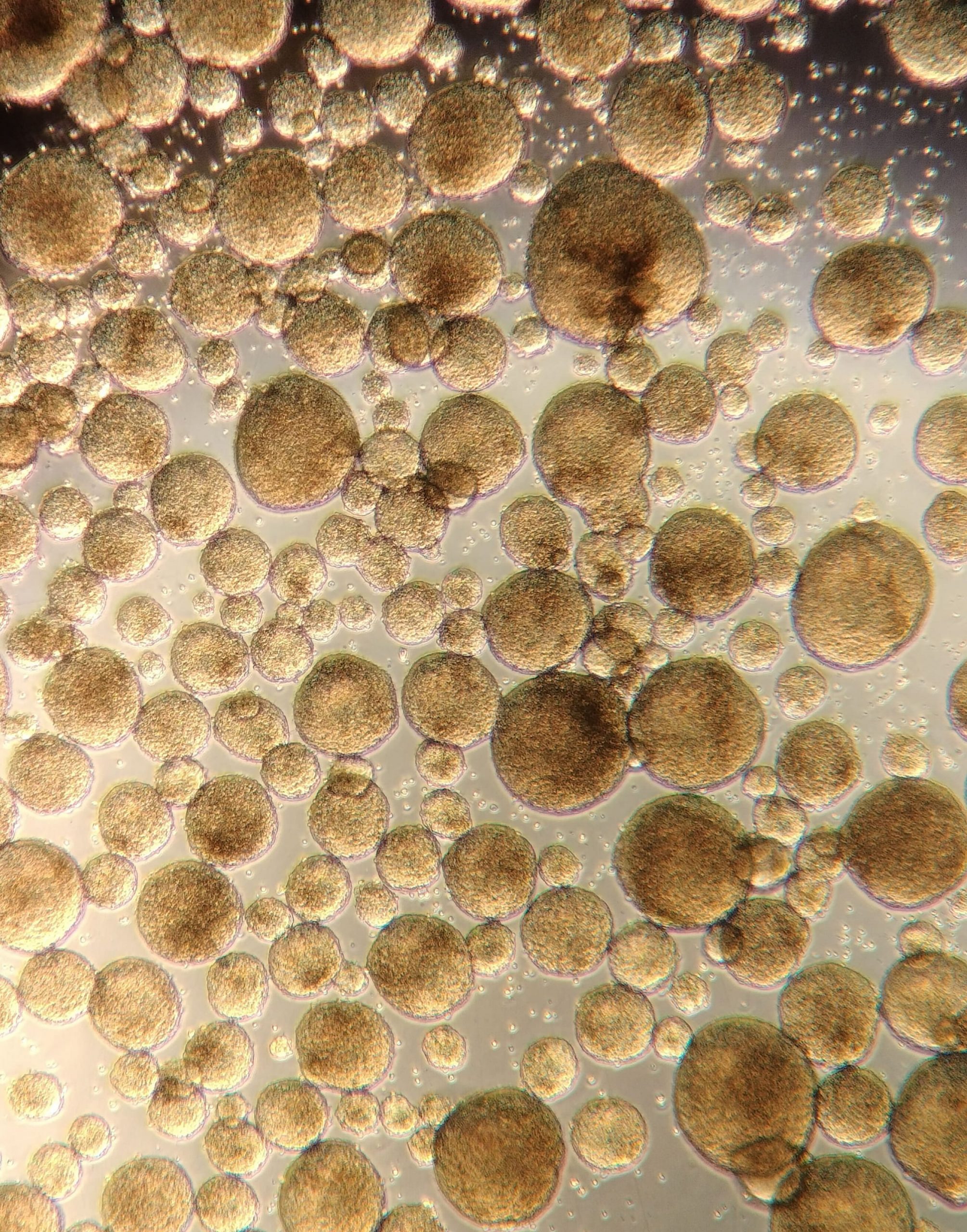
Biomimetic vascular organoids as a model for HHT and PH
We are developing biomimetic 3D vascular organoids derived from patient-specific iPSCs to model HHT and PH. These organoids mimic the architecture of small blood vessels and enable us to study disease-relevant processes such as vascular dilation, intussusceptive angiogenesis, and vessel fragility. This model supports both basic discovery and the future testing of therapeutic interventions.

Artery-on-a-Chip Disease Modeling
Our lab is developing a microfluidic artery-on-a-chip platform to investigate the molecular and cellular mechanisms underlying vascular remodeling in Pulmonary Arterial Hypertension. This device is designed to recapitulate the intimal-medial interface of human arteries by co-culturing endothelial cells and vascular smooth muscle cells, enabling the study of paracrine signaling between these layers during disease progression. This disease model aims to uncover how cell–cell communication, biomechanical forces, and microenvironmental cues contribute to early vascular changes and smooth muscle cell proliferation. We are collaborating with an external bioengineering lab specializing in microfluidic design to fabricate this chip, that supports the development of new therapeutics.
