Research in my lab focuses in three areas:
1) To find effective therapies that can improve pancreatic beta cell preservation and glucose homeostasis in T1D;
2) To uncover therapeutic agents that can induce strong pancreatic beta cell regeneration; and,
3) To understand the intracellular signaling networks regulating the adaptive pancreatic beta cell expansion that occurs in situations of insulin resistance.
The knowledge obtained from these studies can lead to the development of future effective and innovative therapies to treat diabetes.
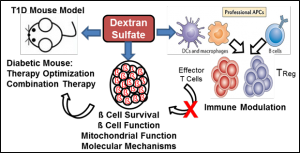 Project 1: Pancreatic Beta Cell Preservation and Immune Regulation in T1D: Using different mouse models of T1D, mouse and human pancreatic islets ex vivo, mouse and human immune cells and the latest state-of-the-art technologies, we are committed to determine whether dextran sulfate reverses T1D by preserving functional pancreatic beta cells in an islet inflammation environment, and by modulating the immune system to enhance tolerance. These studies are funded by NIH/NIDDK and the Department of Defense.
Project 1: Pancreatic Beta Cell Preservation and Immune Regulation in T1D: Using different mouse models of T1D, mouse and human pancreatic islets ex vivo, mouse and human immune cells and the latest state-of-the-art technologies, we are committed to determine whether dextran sulfate reverses T1D by preserving functional pancreatic beta cells in an islet inflammation environment, and by modulating the immune system to enhance tolerance. These studies are funded by NIH/NIDDK and the Department of Defense.
Project 2: Therapeutic Agents to Induce Pancreatic Beta Cell Regeneration. This project analyzes in vivo the beta cell regenerative capacity of (1) harmine, (2) newly generated derivatives of harmine and (3) inhibitors of signaling pathways that repress mitogenic signals in pancreatic beta cells. For these studies, we use three in vivo approaches: (1) administration of these compounds in normal mice, (2) administration in partially pancreatectomized mice, and (3) administration in immunocompromised mice carrying human islets transplanted under the kidney capsule. These studies are funded by NIH/NIDDK and the Juvenile Diabetes Research Foundation and being performed in collaboration with Robert DeVita and Andrew F. Stewart.
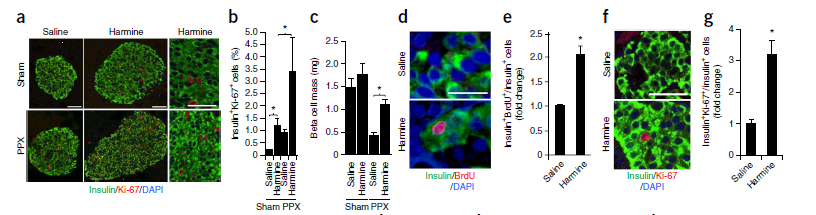
Effect of Harmine in Mouse and Human Beta Cells In Vivo. Nat. Med. 2015; 21(4):383-388.
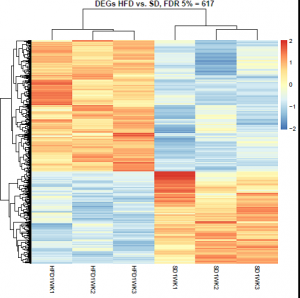
RNASeq Analysis of Islets after High-Fat Diet Feeding
Project 3: Nutrigenomics in the Islet. Does a Chronic Metabolic Stress in Childhood Accelerate the Aging of Young Pancreatic Beta Cells? Childhood obesity and childhood onset T2D are emerging public health issues. Little is known about the molecular mechanisms of the onset of the disease, though epigenetics likely plays an important role. Young beta cells have an open chromatin phenotype with minimal DNA methylation of proliferative and metabolic genes allowing proliferation and adaptive expansion of beta cell mass. Old beta cells display proliferative/metabolic genes that are hypermethylated, with more closed chromatin, correlating with a decreased capacity to proliferate. HFD causes hypermethylation of key hepatic genes. We are testing the hypothesis that a chronic hypercaloric diet phenocopies aging islets, and increases the likelihood of childhood diabetes. These studies are funded by a Pilot and Feasibility Grant from the Mindich Child Health and Development Institute at Icahn School of Medicine at Mount Sinai and being performed in collaboration with Donald K. Scott and Martin Walsh.
Human Islet and Adenovirus Core (HIAC) of the Einstein-Sinai Diabetes Research Center (DRC). My lab is the HIAC that provides training and hands on human islets, isolated rodent islets, islet transplantation approaches and adenovirus and lentivirus generation for transduction of these islets. More information about these services can be found at: https://www.einstein.yu.edu/centers/diabetes-research/human-Islet-and-adenovirus-core/
