Multiple sclerosis is an inflammatory/demyelinating disease of the central nervous system characterized by neurodegenerative processes that occur earlier in life than in classical neurodegenerative diseases. MS aetiology is unknown, and the pathogenesis is believed to be autoimmune. The vast majority of MS patients present with a relapsing-remitting course are characterized by clinical and radiological flares, followed by complete or partial recovery. Currently, more than ten disease-modifying treatments are available, and several others are in different experimental stages. Despite this, there is still a need to identify new therapeutic strategies, effective not only on the inflammatory but also on the degenerative phase of the disease. Our long-term goals are to identify the mechanisms underlying the pathophysiology of MS and to help develop new strategies for an earlier and more accurate diagnosis. Our research tools include neuroimaging methodologies at high- and ultra-high field MRI, optical coherence tomography, behavioral tasks, and electrophysiology.
Our studies are directed towards the identification of MRI biomarkers of disease progression to aid prediction of clinical outcome and treatment choice. Since MS is a chronic, disabling disease that impairs both the physical functions and cognitive ability of patients, we investigate the neural, functional, and structural correlates of neurological deficits. We also seek to determine the functional and structural mechanisms underlying repair and restoration of neurological functions.

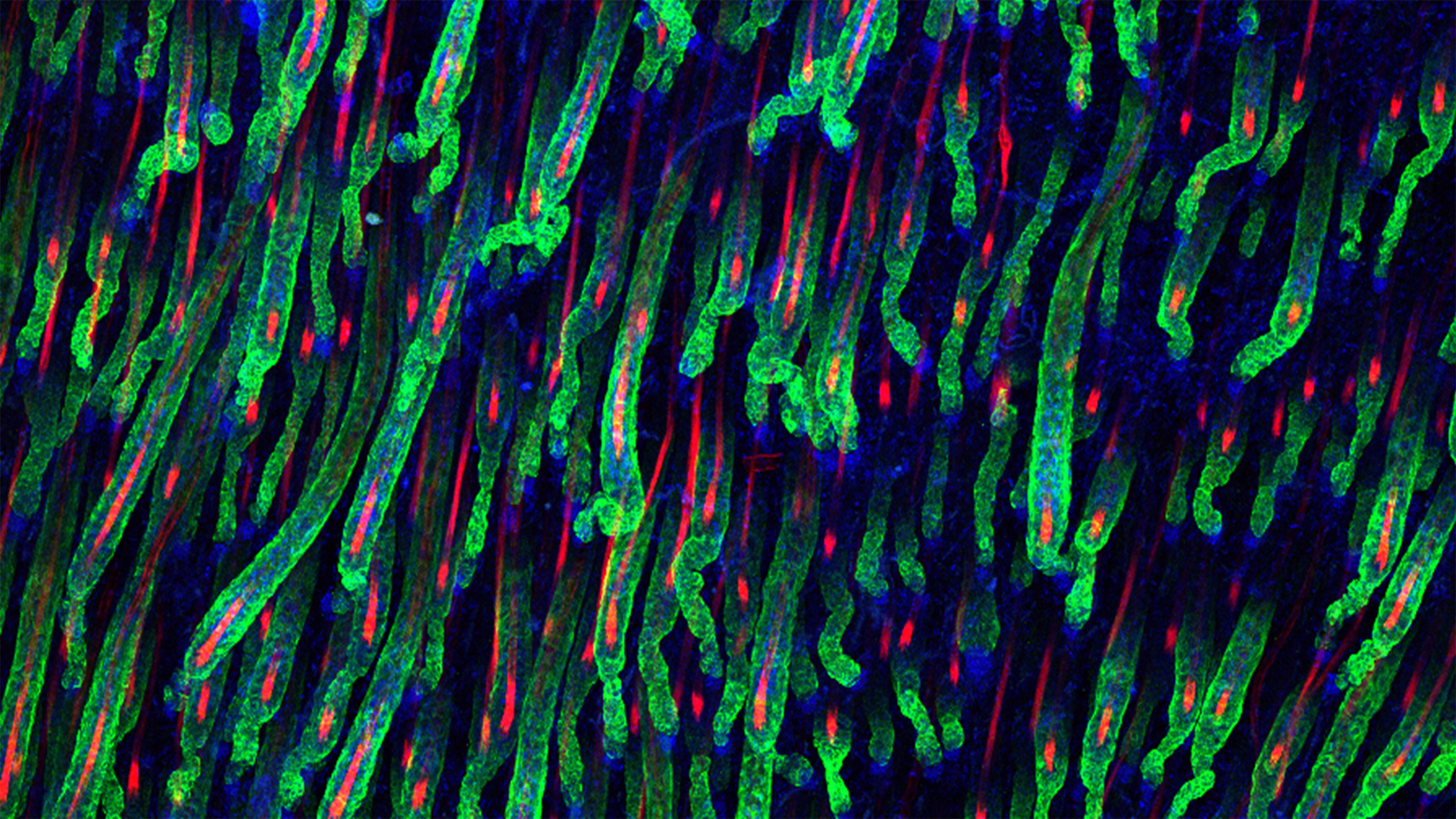
Birth of the Hair Follicle Niche
A central question in stem cell research is how cells specialize during organ development to become adult tissue-regenerative stem cells and their instructive niche. During hair follicle development, epidermal progenitors in nascent hair placodes receive niche signals from specialized cell clusters located underneath that are called Dermal Condensates.
Formation and Function of Stem Cell Niches
Stem cells and their immediate environment, also called stem cell niche, are critical for the growth and continuous renewal of our organs, and are also key for new growth and repair during regeneration. Unlocking the secrets of how specialized niche cells operate to activate stem cells and progenitors holds great promise for regenerative medicine.
Our laboratory aims to answer these fundamental questions in stem cell biology by utilizing the hair follicle mini-organ as a powerful model system. Deciphering how niche cells govern stem cell and progenitor functions in the hair follicle will also provide a rational basis for developing hair regenerative therapies. Fundamental questions towards this goal are:
- How do niche cells acquire the stem cell-inducing properties?
- What are the niche signals that control tissue growth and regeneration?
- How do niche cells control progenitor death and tissue reorganization?
Growth Control and Regeneration
How is tissue growth, homeostasis and renewal by stem cells regulated throughout life? Understanding the core molecular controls is fundamental for realizing the therapeutic potential of the instructive niche (Rezza et al, Current Topics in Developmental Biology 2014). After initial morphogenesis of hair follicles, the Dermal Condensate gives rise to the adult follicle inductive niche, the Dermal Papilla. Dermal Papilla cells signal to progenitors in the follicle bulb to control continuous production and outgrowth of hair. They are also critical for activating stem cells to regenerate a new follicle during the hair growth cycle. Remarkably, isolated niche cells can induce new hair formation after cell transplantation into skin in mice, a yet unrealized goal in human.


Tissue Regression and Reorganization
How tissues regenerate from stem cells and how different organs balance cell loss with new growth is under intensive investigation in the regenerative biology field. In the natural hair growth cycle after years of continuous hair growth, follicles enter a wave of massive progenitor death along the hair axis that – as we are just beginning to unravel – is controlled by the niche microenvironment. During this follicle regression, the Dermal Papilla niche needs to relocate from the base of growing hair bulbs to the stem cell reservoir in the upper follicle for new growth activation.
Tissue Regression and Reorganization
How tissues regenerate from stem cells and how different organs balance cell loss with new growth is under intensive investigation in the regenerative biology field. In the natural hair growth cycle after years of continuous hair growth, follicles enter a wave of massive progenitor death along the hair axis that – as we are just beginning to unravel – is controlled by the niche microenvironment. During this follicle regression, the Dermal Papilla niche needs to relocate from the base of growing hair bulbs to the stem cell reservoir in the upper follicle for new growth activation.